Cristobalite
cristobalite
sanidine
fayalite
tridymite
Images
Formula: SiO2
Simple oxide, tectosilicate (framework silicate)
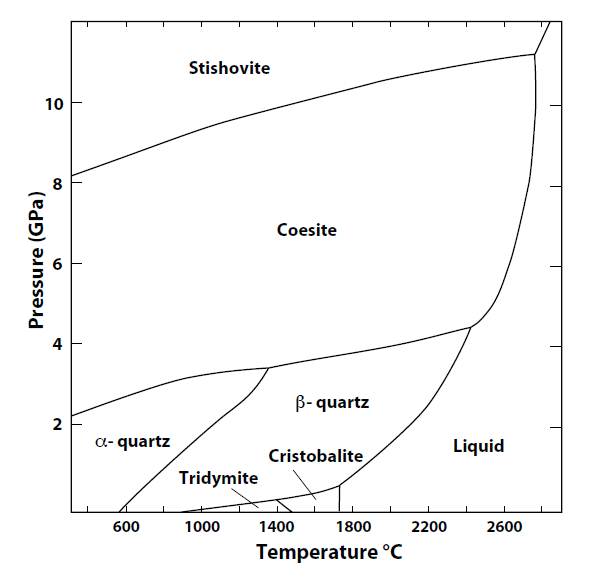
Cristobalite is a paramorph of
quartz
that exists both as α
and as β phases. β-cristobalite is
the stable form of SiO2 from 1,460oC to the melting point, 1,728oC.
It exists as a metastable phase below 1,470oC because the transition to
tridymite proceeds very slowly. The transformation
from β to α-cristobalite occurs at 268oC for pure cristobalite, but may be as low as
175oC if a high level of impurities exists.
Specific gravity: 2.2 to 2.33
Hardness: 6½
Streak: White
Colour: Colourless, white, also blue grey, brown, grey, yellow
Common impurities: Fe,Ca,Al,K,Na,Ti,Mn,Mg,P
Environments:
Volcanic igneous environments
Metamorphic environments
Meteorites
Cristobalite occurs both as α and as β forms as a component of
opal. Crystalline material occurs in silica-rich volcanic
rocks. It is characteristic in vesicles in rhyolite,
andesite and trachyte,
where it
is associated with tridymite,
quartz, sanidine,
pyroxene, fayalite and
magnetite, and also in
basalt.
It is also found in thermally metamorphosed sandstone
(Dana).
Alteration
At atmospheric pressure, with increasing temperature tridymite alters to
cristobalite at 1,470oC, and cristobalite melts at 1,705oC.
tridymite, cristobalite and beta quartz
can co-exist in
equilibrium at a point with temperature about 1,400 oC and pressure 30 kbar
(QP).
Back to Minerals
