Formula: SiO2
Tectosilicate (framework silicate)
Varieties
Ametrine crystals are made of alternating sectors of purple and yellow to orange colour, being amethyst and citrine respectively.
Amethyst is a violet to purple variety of quartz that owes its color to gamma irradiation and the presence of traces of iron built into its crystal lattice.
Binghamite, silkstone and Minnesota tiger's-eye, like many materials denoted by informal lapidary terms, are not well-defined; the distinction between them can be subtle and at times ambiguous; they grade into one another and can be found together on the same large specimen. See entry below for the Cuyuna Range, Minnesota, USA.
Blue quartz is a blue variety of quartz. Some examples of blue quartz are coloured by submicron-size inclusions of ilmenite which produce scattering. Other examples owe their colour to submicron size inclusions of rutile, tourmaline or amphibole, and in rare cases to incorporation of cobalt.
Citrine is a variety of quartz coloured yellow by submicroscopic distribution of colloidal ferric hydroxide and oxides, as well as Fe3+ substituting for Si.
Faden quartz is a group of quartz crystals, usually tabular, with a white thread-like zone running through the interior.
Ferruginous quartz is coloured red-brown by inclusions of hematite.
Green quartz owes its colour to inclusions of chlorite.
Milky quartz is a semi-transparent to opaque white-coloured variety of quartz.
Rose quartz is a variety of quartz which, when massive, is coloured by scattering of tiny orientated rutile needles and/or the presence of Ti3+ in channels and voids. Al3+ is usually also present. Transparent single crystals of rose quartz are coloured by substitutional phosphorus.
Smoky quartz is a variety of quartz coloured dark brown to black due to the presence of Al3+ in the tetrahedral site. If Fe3+ is present in greater concentration than Al3+, irradiation of clear quartz produces smoky quartz at first, but further irradiation further ionises the iron and causes charge transfer between Fe4+ and a trapped hole on an oxygen atom of the Al3+ tetrahedron producing amethyst.
Tiger's eye is a variety of quartz which is chatoyant because of subparallel intergrowth of quartz crystals and altered amphibole fibres that mostly turned into limonite.
Properties of quartz
Crystal System: Trigonal
Specific gravity: 2.65 to 2.66 measured, 2.66 calculated
Hardness: 7
Streak: White
Colour: White or colourless, also grey, yellow, purple, pink, brown, black. Also may be coloured by blue, green or red-brown by inclusions of other minerals.
Solubility of quartz
In water, hydrochloric, sulphuric and nitric acid: quartz is insoluble at atmospheric pressure and temperature, but solubility increases rapidly with pressure and temperature.
In alkalis: SiO2 is attacked by alkaline substances (like potassium hydroxide, KOH). The speed of the reaction depends on the texture and crystal size: crystalline quartz will dissolve only very slowly in hot watery alkaline solutions, while amorphous SiO2, will be readily dissolved at room temperatures, according to the equation:
SiO2 + 2KOH → K2SiO3 + H2O
In hydrofluoric acid: Hydrofluoric acid, HF, will decompose quartz to form first silicon fluoride SiF4, then hydrofluorosilicic acid, H2SiF6:
SiO2 + 4HF → SiF4 + 2H2O
SiF4 + 2HF → H2SiF6
In natron or potash: All forms of silica dissolve in molten natron or potash (K2CO3)
SiO2 + Na2CO3.10H2O → Na2SiO3 + CO2 + 10H2O
SiO2 + K2CO3 → K2SiO3 + CO2
Weathering of silicate rocks: Carbonic acid H2CO3 present, for example, in meteoric waters, releases silicic acid and forms carbonates.
Formation of orthosilicic acid: Quartz dissolves in water at sufficiently high pressure and temperature forming orthosilicic acid, H4SiO4:
SiO2 + 2H2O → H4SiO4
Orthosilicic acid is a very weak acid, weaker than carbonic acid. It dissociates according to the reaction:
H4SiO4 + H2O ⇌ H3SiO4- + H3O+
Environments: Quartz occurs in every type of mineral environment.
In the Bowen reaction series quartz is the last major mineral to crystallise out.
It is the most common mineral found on the surface of the Earth; it occurs in plutonic igneous environments including pegmatites and carbonatites, in sedimentary environments, in contact and regional metamorphic environments, in hydrothermal deposits and it is the principal constituent of hydrothermal veins.
Smoky quartz occurs in alpine fissures and veins; rose quartz occurs in pegmatites.
Quartz is generally a primary, rock-forming mineral, but it may also be of secondary origin.
It is an essential constituent of quartzolite, granite, pegmatites, rhyolite and sandstone.
It is a common constituent of diorite, basalt, phyllite, gneiss and eclogite.
Quartz also may be found in syenite, gabbro, trachyte, andesite, clay, limestone and dolostone.
Quartz occurs in all metamorphic facies with the possible exception of the sanidite facies, where the high temperature paramorph tridymite may occur instead.
Alteration
Quartz is ubiquitous and a component of many reactions which are detailed elsewhere, according to the other components.
The only alterations considered here are transformations between the different paramorphs of SiO2.
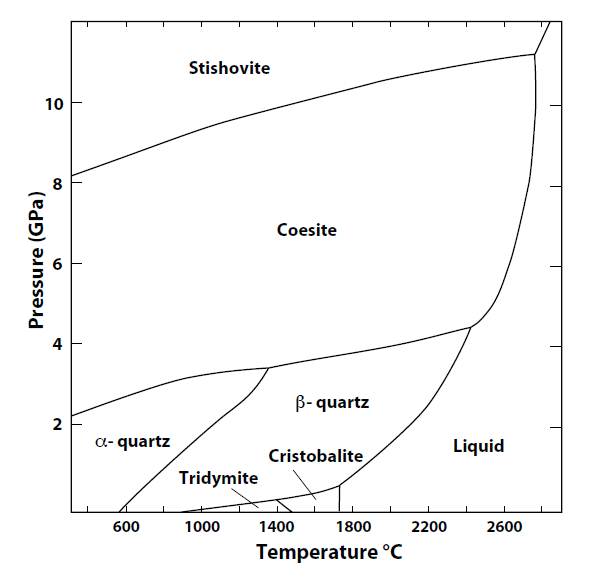
Alpha quartz is the low temperature, low pressure paramorph. At atmospheric pressure it is stable up to 573oC, when it alters to beta quartz, the beta quartz alters to tridymite at 870oC. Alpha quartz, beta quartz and coesite can co-exist at a point where the temperature is about 1,360oC and the pressure 34 kbar.
At atmospheric pressure and 1,470oC tridymite alters to cristobalite, and cristobalite melts at 1705oC.
Tridymite, cristobalite and beta quartz can co-exist in equilibrium at a point with temperature about 1,400 oC and pressure 30 kbar.
Coesite is a high pressure paramorph of quartz. With increasing pressure, at 800oC alpha quartz alters to coesite at about 30 kbar pressure, then coesite alters to the ultra-high pressure paramorph stishovite at about 90 kbar.
beta quartz and coesite can co-exist in equilibrium with the silica melt at a point where the temperature is about 2,410oC and the pressure 45 kbar.
With further increase in pressure and temerature, coesite can continue to exist up to about 2,770oC and 110 kbar pressure, at which point coesite, stishovite and the silica melt are in equilibrium (QP).
The Tyndall Effect in Quartz
The so-called Angel Feather Quartz is caused by the Tyndall Effect.
Watch the video
The Tyndall effect is light scattering by particles in a colloid or in a very fine suspension. Blue light is scattered much more strongly than red light, so it is a blue colour that we see when white light is scattered. The Tyndall effect is seen when light-scattering particulate matter is dispersed in an otherwise light-transmitting medium, where the diameter of an individual particle is in the range of roughly 40 to 900 nm, that is somewhat below or near the wavelengths of visible light (400–750 nm).
For example, blue eyes appear blue due to Tyndall scattering in a translucent layer in the iris. Brown and black irises have the same layer except with more melanin in it, and the melanin absorbs light (Wiki).
The Tyndall effect in quartz arises in rare cases when white light is passed through the crystal and becomes scattered by minute particles, formed during previous growth stages of the crystal. The crystal appears colourless and featureless in normal lighting conditions, but when a narrow beam of white light is passed through it, distinct feathery blue phantom features can be seen. This is particularly dramatic when the quartz has been shaped into a crystal sphere.
Images
Localities for quartz
The Two Mile and Three Mile deposits, Paddy's River, Paddys River District, Australian Capital Territory, Australia, are skarn deposits at the contact between granodiorite and volcanic rocks. Quartz is a primary silicate that occurs in wide veins commonly intergrown with magnetite. Excellent crystals of milky quartz up to 4.5 cm long have been found, some coated with chlorite and magnetite (AJM 22.1.35).
At the Manuka Mine, Mouramba county, New South Wales, Australia, many large smoky quartz crystals were discovered in the copper-lead-silver prospect, and they exhibit a wide variety of forms, and both Dauphine and Japan law twins. The quartz crystals occur in two different lithographies.
The first is in cavities up to 20 cm across in fossiliferous limestone, as crystals that are likely to have formed during a galena-sphalerite-acanthite-quartz hydrothermal mineralisation event.
The second is in leached saprolitic clay overlying dolomitised limestone, in calcium-rich bands within the clay (AJM 22.2.55).
Quartz from the Manuka Mine - Image
At Bahia, Brazil, quartz pseudomorphs after talc have been found (KL p254).
Quartz from Bahia - Image
At the Santa Teresa field, Espírito Santo, Brazil, a kind of quartz occurs that is known locally as "super seven" because it contains seven varieties of quartz: colourless, amethystine, citrine, smoky quartz, rutilated, goethite-included and cacoxenite-included. It is interesting, but not very beautiful (Minrec 54.735).
Quartz from Espírito Santo - Image
At Iraí, Rio Grande do Sul, Brazil, quartz pseudomorphs after calcite have been found (KL p247).
Quartz from Iraí - Image
At the Rock Candy mine, British Columbia, Canada, quartz pseudomorphs after fluorite have been found (KL p251).
At Kitwanga, Skeena, British Columbia, Canada, acicular quartz has been found. This is a very rare habit for quartz, but a road cut near Kitwanga has produced excellent examples of these crystals. They occur in quartz veins cutting through a dark sandstone that is brecciated and extensively replaced by quartz. The acicular crystals are associated with prismatic quartz of normal proportions that are up to 8 mm long, massive calcite and corroded calcite crystals, microcrystals of anatase, brookite, a clay mineral in the kaolinite group, pyrite, and spheres of a black manganese mineral. The acicular quartz was the first mineral to form, and crystals can be found protruding from quartz of normal morphology that grew around them. The acicular quartz crystals are 1 to 3 mm in length and as thin as 0.05 mm or less. Ratios of width to length of 1:50 have been observed. These slender crystals are easily bent; the distortion is elastic and when pressure is released, the crystal springs back to its original form. When the elastic limit is exceeded, the crystal breaks free with great energy and disintegrates. This scale-dependent behaviour is shown by acicular crystals of many minerals such as cuprite, halite and natrolite, but is not often observed (R&M 97.6.566-567).
At the Lyndhurst area, Ontario, Canada, strange crystals of sceptre quartz have been found, often with the cap displaced from the trunk (R&M 97.3.254-259).
The amethyst mines of the Thunder Bay area, Ontario, Canada, are the source of of some large groups of crystals of quartz variety amethyst. Some are of the typical purple amethyst colour, and some have a distinctly red colour, due to sub-surface inclusions of hematite (R&M 94.4.306-341).
At Jinlong Hill, Longchuan County, Heyuan, Guangdong, China, yellow citrine and red hematite-included quartz and colourless quartz have been found (AESS).
Citrine from Jinlong - Image
Hematite-included Quartz from Jinlong
Colourless Quartz from Jinlong
In the vicinity of the Discovery Bay lookout, Lantau Island, Islands District, New Territories, Hong Kong, China, there are several hydrothermal quartz veins and rare vugs with quartz crystals. Some of the massive quartz of the vein shows very pale amethyst colour (Mindat).
At Lantau Peak, Lantau Island, Islands District, New Territories, Hong Kong, China, colourless quartz has been found (AESS).
Quartz from Lantau Peak - Image
At Lin Fa Shan, Tsuen Wan District, New Territories, Hong Kong, China, aggregates of jumbled, poorly formed, translucent white quartz crystals have been found (AESS).
Quartz from Lin Fa Shan - Image
At Castle Peak, Tuen Mun District, New Territories, Hong Kong, China, well formed clear colourless crystals of quartz up to 2.4 cm in length have been found (AESS).
Quartz from Castle Peak - Image
At Kwun Yum Shan, Yuen Long District, New Territories, Hong Kong, China, the deposit is a hydrothermal deposit which lies along a fault zone withi altered acid volcanic rocks, consisting mainly of chlorite, biotite, sericite and actinolite with scattered quartz (Hong Kong Minerals (1991). Peng, C J. Hong Kong Urban Council).
There are several “hot pots” near the top of the hill. These hot pots were thought to be outlets of warm and moist air, which is heated below the ground and ejected through fissures and cracks in the rocks. The rocks here, however, are more likely to be pyroclastic in nature. Mineral veins of quartz, pyrite and galena can be identified, and large crystals of quartz are present in the rock. The Hong Kong Geological Survey has now re-interpreted the rock as an altered intrusive rhyolitic hyaloclastite. It is possible that the outcrop marks a vent feeder of volcanic rocks (Geological Society of Hong Kong newsletter 14.1).
At Lantau Peak, Lantau Island, Islands District, New Territories, Hong Kong, China, quartz veins with small crystals have been found about halfway along the path between Mui Wo and Sunset Peak, at latitude 22.25994 and longitude 113.97927 (Mindat).
Quartz from Lantau Peak - Image
At Tongbei, Fujian province, China, pseudomorphs of opal variety hyalite after quartz have been found with spessartine (KL p259).
Also well formed almost black smoky quartz crystals with orange spessartine (AESS).
Quartz from Tongbei - Image
In Hunan, China, red hematite-included quartz has been found (AESS).
Hematite-included Quartz from Hunan - Image
At Xianghuapu Mine, Xianghualing Sn-polymetallic ore field, Linwu County, Chenzhou, Hunan, China, smoky quartz has been found with albite (AESS and Mindat photo).
Quartz from Xianghuapo - Image
At the Huanggang Fe-Sn deposit, Hexigten Banner, Chifeng City, Inner Mongolia, China, fine quartz specimens have been found, some as stacks of prase-green, vitreous, translucent crystals, and also as pale pink crystals several cm long, of translucent, vitreous quartz, striated across its length, with a dark grey “rose” of what is probably hematite. (AESS).
Green Quartz from Huanggang - Image
Pink Quartz from Huanggang - Image
In Shandong, China, well formed crystals of smoky quartz are associated with microcline and epidote (AESS).
Smoky Quartz from Shandong - Image
At the Ganluo Mine, Ganluo County, Liangshan Yi, Sichuan, China, single, colorless, vitreous crystals of quartz to 33 mm in length have been found (AESS).
Quartz from Ganluo - Image
At Shengou tin mine, Xide County, Liangshan Yi, Sichuan, China, colourless, vitreous, well terminated crystals of quartz up to 2 cm long have been found (AESS).
Quartz from Shengou - Image
From Tibet, China, a 10.3 cm specimen has been found with small colourless vitreous transparent crystals of quartz almost covering a dark branch shaped matrix. There is on patch of pinkish stilbite crystals 12 mm across, and a thin crystal of apophyllite about 1 cm long (AESS).
Quartz from Tibet - Image
At Cabiche, Quípama, Departamento de Boyacá, Colombia, quartz with bright yellow tips coloured by fibrous inclusions of an apparently amorphous mineral have been found. The fibres have not to date (May 2019) been identified unambiguously, but one possibility is halloysite (R&M 94.3.240-247).
Quartz from Cabiche - Image
At Göpfersgrün, Wunsiedel, Wunsiedel im Fichtelgebirge, Upper Franconia, Bavaria, Germany, talc pseudomorphs after quartz have been found (KL p234).
At Idar-Oberstein, Birkenfeld, Rhineland-Palatinate, Germany, quartz pseudomorphs after calcite have been found (R&M 95.3.275).
Quartz from Idar-Oberstein - Image
At Johanngeorgenstadt, Erzgebirgskreis, Saxony, Germany, quartz was found in all veins. In 2005 the first Japan-law twin, 1.3 cm, was found at the Schaar shaft. The quartz - feldspar - tin veins consist mainly of massive milky quartz, but isolated cavities in the veins may be lined by quartz crystals, some reaching several centimeters in length. Massive milky quartz is commonly shot through with black acicular crystals of schorl and also carries cassiterite. The weakly developed quartz - sulphide ore veins consist of massive, milky white quartz, in which are intergrown chiefly galena with a little pyrite and sphalerite. The veins also showed chert-like quartz; in the silver ore veins mined in the 18th century, chert was noted quite commonly, and in it were intergrowths of silver, plumose bismuth, uranium ores and cobalt - nickel ores. The numerous iron ore veins commonly contained large masses of chert and of jasper in various colours. (MinRec 55.5.617-618).
Quartz from Johanngeorgenstadt - Image
At the Porretta quartz sites, Porretta Terme, Alto Reno Terme, Metropolitan City of Bologna, Emilia-Romagna, Italy, the geological setting is deep marine turbiditic sequences characterised by a rhythmic alternation of sandstone, siltstone, clay and marly beds.
Fractures in the sandstone are characterised by the presence of calcite and quartz crystals. The quartz crystals typically show skeletal growth forms and have inclusions of light yellow-grey clay. They frequently also contain water and gaseous hydrocarbon inclusions. The crystals have smooth and lustrous, in part curved, faces and often form aggregates of two or more crystals, usually in parallel growth. The quartz crystals are found everywhere in rock clefts of the Porretta sandstone formation.
In window quartz (skeleton quartz) the edges grew more quickly than the faces, so the edges stand out like the frames of a window. Crystals that grow very quickly often develop these skeletal growth forms; other examples are gold and halite crystals. The faces on a skeleton quartz crystal grow from the edges to the centre. Sometimes these faces grow as thin transparent plates, and if these plates are finally completed, the watery solution inside will be trapped behind a "window".
Window quartz crystals from this locality are considered to be amongst the best in the world (Mindat, QP, AJM 22.2.13-25).
At the Monte Cervandone area, Devero Alp, Baceno, Verbano-Cusio-Ossola Province, Piedmont, Italy, colourless and smoky prismatic quartz crystals, many of the Tessin habit, can reach up to 65 cm long. Rare quartz crystals with inclusions of acicular rutile and tourmaline, and some showing hollow internal shapes of vanished anhydrite crystals, are highly prized. Collecting sites in the Chummibort drainage have famously yielded crystals of colourless quartz overgrown with a later generation of transparent, medium-purple amethyst; beautiful specimens of sceptre amethyst from Chummibort have been known since around 1900 (MinRec 56.3.318).
Quartz from Chummibort - Image
At Charcas, Charcas Municipality, San Luis Potosí, Mexico, the primary minerals are sphalerite, galena, chalcopyrite, bornite, tetrahedrite, arsenopyrite, pyrite and silver minerals such as jalpaite, diaphorite and acanthite. In the host rock, as metamorphic or alteration minerals, danburite, datolite, hedenbergite, epidote, chlorite, andradite, actinolite and wollastonite have been reported.
Quartz, calcite and danburite crystallised during the entire life of the systems, throughout the intrusive emplacement, metamorphism, and mineralising events. With depth, both sphalerite and galena decrease while chalcopyrite increases.
Secondary sulphides formed include bornite, covellite, digenite and chalcocite. Native silver, native gold, hematite and goethite were deposited after the sulphides (Minrec 55.6.727-728).
Prismatic, hexagonal, colorless and transparent to opaque milky quartz occurred early. Crystals can reach 10 cm in length, and are associated with sphalerite, danburite, datolite or calcite. Some sceptres up to 15 cm have been found. This early quartz can be covered by a late-stage druse of fine rock crystal, citrine and smoky quartz. Skeletal quartz is common; colourless crystals to 25 cm are known. Fenster quartz crystals display fluid inclusions up to 3 mm in size. Japan-law twinning is rare; the few such twins that have been found are translucent, colourless, and reach up to 5 cm in length.
Amethyst is moderately common; crystals can reach a metre in length; specimens collected from the San Bartolo mine show long, greyish purple crystals embedded in golden yellow calcite. Also from the San Bartolo mine are short, stubby, glassy and transparent crystals of amethyst to 1 cm that crystallised on dog-tooth calcite. A similar example consists of 4 mm amethyst crystals coating datolite. Doubly terminated crystals of amethyst to 2 cm perched on colourless white danburite dusted with pyrite have been found, and in 1996, some Japan-law twinned amethyst crystals dusted with pyrite were discovered.
Amethyst from Charcas - Image
Citrine apparently crystallised relatively late in the sequence, yellow grading into amber in colour. Stubby, doubly terminated crystals to 6 cm are moderately abundant, but the majority are under 1 cm. Drusy citrine is not uncommon and coats earlier quartz and danburite. It is very commonly associated with danburite and calcite.
Citrine from Charcas - Image
Ametrine crystals are comparatively rare; they have a good lustre and can reach 3 cm in length.
Rare, prismatic smoky quartz crystals to 3 cm have been found, attached to danburite. Drusy black quartz is sometimes seen coating older quartz crystals.
Examples of epimorphs are common at Charcas, such as fine drusy quartz or citrine after danburite, calcite or earlier generation quartz. The common pseudomorphs are quartz after danburite and calcite after danburite.
Quartz epimorphs after danburite - Image
The occurrence of high-temperature quartz-beta crystals exhibiting the hexagonal-bipyramidal habit and reaching less than 1 cm has been reported (Minrec 55.6.727-728).
At the Berg Aukas mine, Grootfontein, Otjozondjupa Region, Namibia, quartz is a monor constituent in both the country rock and the orebodies. Drusy quartz is most commonly associated with descloizite and, together with calcite, dolomite, goethite and willemite, can form the matrix of some descloizite crystals (R&M 96.2.132).
At the Nikolaevskiy mine, Dalnegorsk, Russia, quartz pseudomorphs after beta-quartz have been found. Also at Dalnegorsk, quartz pseudomorphs after fluorite have been found (KL p246, 252).
At Mkobola district, Mpumalanga province, South Africa, a chalcedony pseudomorph after a quartz Japanese twin has been found (KL p258).
At the Witwatersrand Goldfield, South Africa, quartz is the most common mineral, constituting the bulk of the quartzite and the matrix of the conglomerate; it is also the most common mineral forming the conglomerate clasts. The latter are primarily white to grey vein quartz pebbles although less common quartzite and chert pebbles also occur. Quartz is found associated with baryte, calcite, chalcopyrite, clinochlore, dolomite, epidote, galena, gold, pyrite, pyrobitumen, pyrochlore and pyrrhotite. Several gold mines have produced aesthetic quartz crystals, typically transparent (R&M 96.4.341-342).
At the Welkom goldfield, Lejweleputswa District, Free State, South Africa, large specimens of quartz weighing several kilograms have been found (R&M 96.4.341-342).
At the President Brand Mine, Lejweleputswa District, Free State, South Africa, quartz crystals are partially to wholly coated by a thin film of light green clinochlore producing a glittering sheen. Clinochlore can also be included within some crystals imparting an attractive green colour to the quartz. Doubly terminated crystals have also been found here (R&M 96.4.341-342).
At the Mponeng Mine, West Wits, Far West Rand, West Rand District Municipality, Gauteng, South Africa, well formed quartz crystals to 30 cm, often as doubly terminated floaters, were common (R&M 96.4.341-342).
At the Kusasalethu Mine, Carletonville, Western Sector, Far West Rand, West Rand District Municipality, Gauteng, South Africa, noteworthy transparent crystals of quartz have been found (R&M 96.4.341-342).
At the Kopanang Mine, Klerksdorp, Dr Kenneth Kaunda District Municipality, North West, South Africa, a fault was intersected that yielded quartz specimens that are typically elongated prisms, transparent, and associated with brown, goethite-altered pyrrhotite and pyrite. Quartz specimens associated with grey rhombohedral calcite display distinct growth features via internal zoning, producing phantom forms (R&M 96.4.341-342).
At the Buffelsfontein gold mine, Stilfontein, Dr Kenneth Kaunda District, North West, South Africa, noteworthy transparent crystals of quartz have been found (R&M 96.4.341-342).
In the Berbes mining area, Ribadesella, Asturias, Spain, La Cabaña area is known for some exceptional doubly terminated quartz crystals, similar to the famous “Herkimer diamond” quartz from New York. At Berbes, these crystals are found only in certain regions in La Cabaña, in solution cavities in the host limestone. Crystal sizes vary from less than a millimeter up to 20 cm long, but the average size is between 1 and 3 cm. The crystals are of simple habit, with short prism faces and well-developed rhombohedron faces; sceptres and reverse sceptres are rare. Clusters with crystals radiating in every direction are common; most of these are floaters, with no point of attachment to matrix. Quartz crystals are typically found in zones where the host rock contains high amounts of organic matter or hydrocarbons. Commonly these impurities are trapped as solid, gas and fluid inclusions in the crystals during crystal growth. The inclusions show intense yellow-green fluorescence (MinRec 55.1.68-71).
At Malmberget, Gällivare, Norrbotten County, Sweden, quartz is sometimes found with fluorite, calcite, stilbite-stellerite, fluorapatite, hematite and other species (MinRec 56.5.598).
Quartz from Malmberget - Image
At Asar hill, Güğtı, Dursunbey district, Balikesir province, Marmara region, Turkey, quartz pseudomorphs after calcite have been found (KL p248).
At Wheal Mary Ann, Menheniot, Cornwall, England, UK, quartz pseudomorphs after fluorite have been found (R&M 95.3.275).
In a cavity at the Smallcleugh Mine, Nenthead, Alston Moor, Eden, Cumbria, England, UK, quartz, as the first mineral to form, commonly encrusts fragments of rock, and also occurs as thin pure laths; individual crystals are pyramidal and rarely exceed 0.2 mm across (JRS 18.20).
Examples of quartz epimorphs after fluorite are sparingly present on the mine dumps. Some collected specimens exhibit overgrowths of minute colourless pyramidal quartz crystals, and more rarely sphalerite and pyrite, on the inner surfaces of these epimorphs (JRS 2.97).
Quartz from Smallcleugh - Image
At the Carrock Mine, Mungrisdale, Eden, Cumbria, England, UK, colourless quartz crystals occur with chlorite and pyrite inclusions (AESS).
Quartz from Carrock Mine - Image
At the Fall Hill quarry, Ashover, Derbyshire, England, UK, quartz crystals occur on fluorite (RES p104).
At Calton Hill quarry, Buxton, Derbyshire, England, UK, quartz variety amethyst has been found lining a cavity in basalt (RES p116).
At Diamond Hill, Buxton, Derbyshire, England, UK, quartz variety eisenkiesel occurs in a baryte matrix (RES p134).
At Waterswallows quarry, Buxton, Derbyshire, England, UK, quartz variety amethyst has been found in a vug in dolerite (RES p135).
At Kenslow Knoll sandpit, Newhaven, Derbyshire, England, UK, quartz crystals occur on baryte (RES p114).
At the Harry Stoke mine, near Filton, South Gloucestershire, England, UK, quartz variety amethyst has been found with celestine and calcite (RES p168).
At Croft Quarry, Croft, Blaby, Leicestershire, England, UK, quartz is associated with analcime and is often well crystallised and showing left-hand faces. A cavity has been discovered with walls lined by a film of epidote with a little hematite, upon which was deposited a comb structure of thousands of small quartz crystals up to 12 mm in length and up to 5 mm in cross section. Of 52 crystals examined, 43 showed the development of the left trigonal pyramid {2111}, and rarely the left trigonal trapezohedron {6111} (JRS 20.24-25).
At the Cloud Hill quarry, Breedon on the Hill, Leicestershire, England, UK, quartz crystals have been found with hematite in a cavity in goethite, and also as variety eisenkiesel on a goethite matrix (RES p206).
At the Bardon Hill quarry, Coalville, Leicestershire, England, UK, quartz crystals have been found encrusted with dolomite (RES p194).
At Peldar Tor quarry, Spring Hill, Whitwick, Leicestershire, England, UK, quartz crystals have been found heavily included with chlorite (RES p201).
At the Snailbeach mine, near Minsterley, Shropshire, England, UK, quartz crystals coat both calcite rhombohedra and galena crystals. Quartz also occurs here with cerussite (RES p270, 272, 275, 276).
At the Edwin Richards quarry, Rowley Regis, Dudley, West Midlands, England, UK, quartz variety amethyst has been found with dolomite in a calcite vein (RES p330).
At the Finch mine, Hayden, Banner district, Gila county, Arizona, USA, quartz pseudomorphs after wulfenite have been found (KL p253).
At Black Canyon City, Maricopa county, Arizona, USA, large pseudomorphs of quartz after both anhydrite and aragonite have been found (R&M 94.2.166-167, KL p245).
At the Potter-Cramer mine, Vulture Mining District, Maricopa county, Arizona, USA, secondary colourless quartz crystals fill vugs and fractures within the host rock. Surprisingly the quartz fluoresces bright bluish white under SWUV, but not MW or LW, and it does not exhibit phosphorescence. It is not known what activates the fluorescence (R&M 96.1.34).
At the Holland Mine, Duquesne Gulch, Nash Mines group, Duquesne-Washington Camp, Patagonia Mining District, Patagonia Mountains, Santa Cruz County, Arizona, USA, high-quality Japan-law quartz twins, some very large, have long been known
Quartz from the Holland Mine - Image
At the South Belmont Mine, Santa Cruz county, Arizona, USA, fine epimorphs of quartz after calcite have been found (R&M 94.2.160).
In the South Comobabi Mountains, Pima county, Arizona, USA, epimorphs of quartz after calcite have been found, sometimes with hematite inclusions (R&M 94.2.161).
At the Emmons pegmatite, Greenwood, Oxford county, Maine, USA, quartz can occur as crystals up to 25 cm long. Very dark smoky quartz crystals tend to occur in late-stage secondary phosphate assemblages with rhodochrosite. The Emmons pegmatite is an example of a highly evolved boron-lithium-cesium-tantalum enriched pegmatite (R&M 94.6.514).
The Central Mine, Central, Keweenaw county, Michigan, USA, initially targeted a series of sub-parallel mineralised fissure veins where the most copper-rich portion of the vein was close to the base of the main greenstone flow.
Quartz was an abundant gangue mineral in the fissure veins of the Central mine, and where the vein was vuggy or brecciated, fine crystallized quartz specimens were sometimes encountered, showing colourless and transparent, prismatic crystals up to 8 cm. Usually these quartz crystals are associated with lime-green prehnite, epidote, pumpellyite, copper and rarely silver. More commonly, quartz occurs as stubby 2 to 10 mm prismatic crystals which fill narrow open fractures in basalt, or coat the walls of mineralised amgydules in the basalt flows. (MinRec 54.1.80-81)
At the Connecticut Mine, Delaware, Keweenaw county, Michigan, USA, microscopic quartz pseudomorphs after microcline have been found (R&M 97.4.358).
The Cliff Mine, Phoenix, Keweenaw county, Michigan, USA, is situated at the base of a roughly 70-metre basalt cliff. A curious feature of the impressive thickness of the greenstone flow here is that it contains zones of “pegmatoid”: areas where slow cooling in the core of the lava flow allowed for large feldspar crystals exceeding 1 cm to grow. Such features are normally only observed in intrusive igneous rocks and are almost unheard of in basalt flows.
The Cliff mine primarily exploited rich copper mineralisation in the Cliff fissure (vein). Although mineralised with copper to some extent along its entire length, the part of the vein just below the greenstone flow carried the richest copper mineralisation by far. A significant amount of the copper recovered at the Cliff mine came from amygdaloids in the tops of 13 basalt flows which were cut by the Cliff vein. The discovery and mining of this vein proved that the veins were the source of the large masses of float copper that were already well known, and proved that the primary ore mineral in the district was native copper, not sulphides, as had been suspected earlier.
Quartz as white or colourless and transparent crystals is common in the Cliff vein. Fine examples in association with prehnite and other minerals are known. As for all colourless species occurring at the Cliff mine, copper inclusions in quartz crystals are well documented (MinRec 54.1.25-49).
At the Cuyuna Range, Minnesota, USA, lapidary materials binghamite, silkstone, and Minnesota tiger's-eye are found. They are colorful, often chatoyant, materials formed in banded iron formations, and best defined as types of jasper, but with inclusions of many minerals such as hematite, goethite, siderite, stilpnomelane, minnesotaite, manganese oxides and various amphiboles. All are brightly coloured, generally red, yellow, orange and rich brown mixed together; blue and green varieties are rare. The silica is generally microcrystalline, but younger, macrocrystalline, white quartz also occurs, as do local veins of agate. All contain inclusions of fibrous amphiboles, most of which were pseudomorphically replaced by iron oxides and hydroxides. The amphiboles are mostly crocidolite and grunerite. These are intergrown with the quartz and account for the chatoyancy. (MinRec 56.4.482-486).
Binghamite from the Cuyuna Range - Image
At the Little Gem amethyst mine, Jefferson county, Montana, USA, quartz occurs in a pegmatite outcrop hosted in biotite-hornblende granite and containing pockets of significant size, mostly enclosed partially or completely within microcline feldspar. Pockets in the core of the pegmatite contain only quartz. Elsewhere they contain quartz and microcline, but seldom albite, with occasional minor quantities of epidote, schorl, anatase, pyrite (typically altered to limonite) and goethite.
Quartz occurs in a number of varieties, including milky quartz, rock crystal, smoky quartz and amethyst. Amethyst is a late stage generation of quartz, often the last, exhibiting habits including sceptres and orientated overgrowths. The purple colour is caused by trace quantities of iron as Fe3+, so where there is insufficient iron rock crystal or smoky quartz form in preference to amethyst.
Sceptres are very common, generally with milky or smoky stems and amethyst heads.
Jacaré or crocodile quartz has overgrowths that crystallised from the hydrothermal fluids that created secondary minerals. The substrate is milky to smoky quartz, and the jacaré is usually amethyst.
Many of the quartz crystals have fluid inclusions (R&M 93.6.498-516).
At the PC Mine, Cataract Mining District, Jefferson county, Montana, USA, quartz occurred as single crystals, groups, and as Japan-law twins. Specimens are mostly colourless, rarely smoky. Individual crystals are up to 30 cm long and groups up to 76 cm. Inclusions include anhydrite, hematite, sericite, schorl and pyrite. It is estimated that more than four thousand specimens of Japan-law twins were found here.
A twin is the symmetrical intergrowth of two crystals. A Japan-law twin is a contact twin with the c-axes inclined to each other at an angle of 84.55 degrees. A Reichenstein-Grieserntal twin is a rare contact twin with the inclination of the c-axes at 76.43 degrees. Only five are known to have been found at the PC mine (R&M 96.490-501)
In Nebraska, USA, quartz pseudomorphs after gypsum have been found (KL p249).
At Cookes Peak mining district, Luna county, New Mexico, USA, quartz occurs as massive replacement of limestone bodies, and as crystals lining cavities in these bodies. It is occasionally associated with fluorite, and is sometimes found as quartz epimorphs after fluorite. Excellent epimorphs of quartz after calcite have also been found, some associated with fluorite (R&M 94.3.234-235).
At the Dafoe property, Pierrepont, St. Lawrence county, New York, USA, quartz occurs as primary, Tessin-habit crystals to 15 cm and is found in association with tourmaline. Crystals are normally white to grey, poorly formed, and often coated with white talc. Secondary, or late-stage, quartz occurs as fine, prismatic, transparent crystals to 18 cm. It also is found as attractive molds after calcite, tourmaline and an undetermined species, likely baryte (R&M 97.3.250).
At Rose Road, Pitcairn, St. Lawrence county, New York State, USA, quartz occurs at the skarn deposit as pseudomorphs after wollastonite, either as isolated crystals in areas of coarsely crystallised calcite or as crystals lining the walls of a diopside-albite rock that faces into coarsely crystallised calcite (R&M 97.5.434-444).
At the Devil's Den locality, Mount Tabor, Rutland county, Vermont, USA, collecting is no longer permitted, but in the past it has been a prolific source of various varieties of quartz, including faden quartz, smoky quartz and Mount Tabour "twins". The "twins" are pairs of parallel crystals that share a common prism face and a pair of terminations with a church-and-steeple appearance (R&M 94.3.266-272).
Back to Minerals