In contact metamorphic environments it may be associated with molybdenite.
In hypothermal (high temperature) and mesothermal (moderate temperature) veins and replacement deposits it occurs associated with galena, sphalerite and dolomite. It may contain gold or silver. Chalcopyrite is often present in large bodies of pyrite. Primary chalcopyrite readily alters to the secondary minerals bornite, covellite and brochantite, and also malachite, azurite, langite and numerous other secondary copper minerals.
Localities
The Two Mile and Three Mile deposits, Paddy's River, Paddys River District, Australian Capital Territory, Australia, are skarn deposits at the contact between granodiorite and volcanic rocks. Chalcopyrite is a the main primary copper mineral in the Two Mile deposit, occurring in aggregates and veins with pyrite and magnetite. Chalcopyrite-rich masses up to 70 g containing magnetite, sphalerite, galena and pyrite, have been observed in chloritic skarn. Some chalcopyrite-pyrite masses in hedenbergite skarn contain scheelite enclosed by chalcopyrite (AJM 22.1.36).
At the Mount Kelly deposit, Gunpowder District, Queensland, Australia, the deposit has been mined for oxide and supergene copper ores, predominantly malachite, azurite and chrysocolla. The ores overlie primary zone mineralisation consisting of quartz-dolomite-sulphide veins hosted in dolomite-bearing siltstone and graphitic schist.
Chalcopyrite is the major constituent of the primary ore. It occurs in veinlets associated with pyrite, in carbonate-quartz veins and finely disseminated with pyrite and minor sphalerite in the host siltstone. It also occurs rarely as crystals to 2 mm in quartz veins associated with hematite and partially coated by bornite. Paragenesis for the primary zone is dolomite followed by pyrite, then chalcopyrite and sphalerite, and lastly bornite (AJM 22.1.20 & 25).
At the Mount Lyell Mines, Queenstown, Queenstown district, West Coast municipality, Tasmania, Australia, chalcopyrite is associated with chamosite (Mindat photo).
Chalcopyrite from the Mount Lyell Mines - Image
At Mount Moliagul, Moliagul, Central Goldfields Shire, Victoria, Australia, chalcopyrite is present as coatings on pyrite crystals in granodiorite and as anhedral masses in quartz veins (AJM 21.1.42).
In Bulgaria pyrite pseudomorphs after chalcopyrite have been found (R&M 95.3.275).
Chalcopyrite from Bulgaria - Image
At the Blue Point mine, Thunder Bay, Ontario, Canada, chalcopyrite is associated with galena, and minor pyrite and marcasite (R&M 94.4.319).
Chalcopyrite from Blue Point - Image
At the Thunder Bay Amethyst Mine, Thunder Bay District, Ontario, Canada, chalcopyrite has been found as inclusions in quartz variety amethyst (R&M 94.4.332).
At the Xiling Calcite Mine, Gongcheng County, Guilin, Guangxi, China, chalcopyrite has been found as a dusting of tiny crystals on matrix (AESS).
Chalcopyrite from Xiling - Image
At the Lingchuan Mine, Lingchuan County, Guilin, Guangxi, China, a specimen has been found with dark brassy
Chalcopyrite from Lingchuan - Image
At the Lin Ma Hang mine, North District, New Territories, Hong Kong, China, the lead-zinc deposit is a hydrothermal deposit which lies along a fault zone within altered acid volcanic rocks, consisting mainly of chlorite, biotite, sericite and actinolite, with scattered quartz. (Hong Kong Minerals (1991). Peng, C J. Hong Kong Urban Council)
The mineralisation consists of a series of fissure vein deposits varying from a few mm to several metres on width. The initial vein filling was coarse milky quartz. this was followed by an intrusion of fine-grained quartz carrying the metallic minerals, galena, pyrite, sphalerite and chalcopyrite, in order of abundance (Geological Society of Hong Kong Newsletter, 9.4.3-27).
Malachite and azurite are occasionally found in close association with chalcopyrite (Hong Kong Minerals (1991). Peng, C J. Hong Kong Urban Council).
At Devil's Peak, Sai Kung District, New Territories, Hong Kong, China, the mineralisation occurred in quartz veins in the contact zone between a granite intrusion and acid volcanic rocks. The mine is now closed, and inaccessible for collecting. Chalcopyrite occurred in the quartz veins with beryl, wolframite, molybdenite, pyrite and arsenopyrite (Hong Kong Minerals (1991). Peng, C J. Hong Kong Urban Council)
The Ma On Shan Mine, Ma On Shan, Sha Tin District, New Territories, Hong Kong, China, is an abandoned iron mine, with both underground and open cast workings. The iron ores contain magnetite as the ore mineral and occur predominantly as masses of all sizes enclosed in a large skarn body formed by contact metasomatism of dolomitic limestone at the margins of a granite intrusion. In parts of the underground workings magnetite is also found in marble in contact with the granite. The skarn rocks consist mainly of tremolite, actinolite, diopside and garnet. Malachite and azurite are occasionally found in close association with chalcopyrite, formed by the action of carbonated water on the chalcopyrite (Hong Kong Minerals (1991). Peng, C J. Hong Kong Urban Council)
The Needle Hill Mine, Needle Hill, Sha Tin District, New Territories, Hong Kong, China, is a tungsten mine, abandoned in 1967. The principal ore is wolframite, and the principal gangue mineral is quartz. Molybdenum also occurs. The mineralisation consists of a series of parallel fissure veins that cut through granite. Wolframite and quartz are the main minerals, but galena, sphalerite, pyrite, molybdenite and fluorite have also been found here (Geological Society of Hong Kong Newsletter 9.3.29-40). The quartz-wolframite veins are of high-temperature hydrothermal formation, and grade into wolframite-bearing pegmatites.
Chalcopyrite is sometimes associated with wolframite (Hong Kong Minerals (1991). Peng, C J. Hong Kong Urban Council)
At Chuen Lung, Tsuen Wan District, New Territories, Hong Kong, China, in fissure veins in granite rocks in a small stream near Chuen Lung, silver-bearing galena occurs associated with massive granular amber coloured sphalerite, chalcopyrite, pyrite and pyrrhotite (Hong Kong Minerals (1991). Peng, C J. Hong Kong Urban Council)
The Lin Fa Shan deposit, Tsuen Wan District, New Territories, Hong Kong, China, is located in a remote area of the Tai Mo Shan Country Park, on a steep west facing slope of Lin Fa Shan, just above the abandoned village of Sheung Tong. The surrounding hillsides are covered with shallow excavations, representing past searches for wolframite, the natural ore of tungsten. The abandoned workings are extremely dangerous with unsupported tunnels, open shafts and no maintenance since their closures in 1957; the workings should not be entered (http://industrialhistoryhk.org/lin-shan).
Malachite and azurite are occasionally found in close association with chalcopyrite formed by the action of carbonated waters on the chalcopyrite (Hong Kong Minerals (1991). Peng, C J. Hong Kong Urban Council).
At the Chashan Mine, Xianghualing Sn-polymetallic ore field, Linwu County, Chenzhou, Hunan, China, chalcopyrite has been found as small crystals in granite (AESS).
Chalcopyrite from Chashan - Image
At Manaoshan Mine, Dongpo ore field, Suxian District, Chenzhou, Hunan, China, massive chalcopyrite has been found (AESS).
Chalcopyrite from Manaoshan - Image
In Jiangxi, China, some fine specimens of chalcopyrite have been found, including some that are irridescent due to interference colours (AESS).
Chalcopyrite from Jiangxi - Image
At the Chipuyuguang Mine, Chipu Pb-Zn deposit, Ganluo County, Liangshan Yi, Sichuan, China, brassy yellow chalcopyrite occurs with dark brownish sphalerite (AESS).
At the Shengou lead-zinc mine, Xide County, Liangshan Yi, Sichuan, China, brassy yellow chalcopyrite has been found with black magnetite (AESS).
At Johanngeorgenstadt, Erzgebirgskreis, Saxony, Germany, chalcopyrite was widespread but sparse in the bismuth - cobalt - nickel veins. Concentrations of chalcopyrite were found sporadically in the following mines: Gnade Gottes, Römisch Adler, Georg Wagsfort, Samuelis, Gottes Segen and Neu Leipziger Glück. In the Neujahr mine, crystal druses of iridescent chalcopyrite were found, and in the Eleonora mine it occurred as botryoidal formations with crystallized surfaces. Occasionally chalcopyrite was found in gravel beds (MinRec 55.5.591).
At Herrensegen, Schapbach, Schartzwald, Germany, chalcopyrite occurs with covellite (FM 2318).
At Lettermuckoo Quarry, Kinvarra, Connemara, Galway County, Connacht, Ireland, the minerals are hosted by a megacrystic pink to grey monzogranite, with occasional pegmatitic segregations.
Tiny (<1 mm) golden crystals of chalcopyrite occasionally overgrow fluorite and baryte. Rarely, isolated sphenoidal crystals and crystal groups, which may reach several millimetres across, occur (JRS 22.40).
At Guanajuato, Mexico, acanthite-chalcopyrite pseudomorphs after polybasite have been found (KL p124).
At Charcas, Charcas Municipality, San Luis Potosí, Mexico, the primary minerals are sphalerite, galena, chalcopyrite, bornite, tetrahedrite, arsenopyrite, pyrite and silver minerals such as jalpaite, diaphorite and acanthite. In the host rock, as metamorphic or alteration minerals, danburite, datolite, hedenbergite, epidote, chlorite, andradite, actinolite and wollastonite have been reported.
Quartz, calcite and danburite crystallised during the entire life of the systems, throughout the intrusive emplacement, metamorphism, and mineralising events. With depth, both sphalerite and galena decrease while chalcopyrite increases.
Secondary sulphides formed include bornite, covellite, digenite and chalcocite. Native silver, native gold, hematite and goethite were deposited after the sulphides (Minrec 55.6.727-728).
Chalcopyrite occurs mostly as microscopic inclusions or blebs, but macrocrystals do occur. Crystals are most commonly small, measuring 2 to 4 mm, and occur sprinkled on or included in danburite, datolite and early-formed quartz crystals. Crystals of chalcopyrite to 3 cm in size are gold-coloured, unless tarnished to an iridescent purple colour, and are found scattered on a skarn or vein matrix such as sugary quartz or on small individual quartz crystals with calcite. Chalcopyrite is associated with other sulphides such as pyrite, galena and sphalerite. In 2002 clusters of pseudo-octahedral to pseudocubic chalcopyrite crystals up to 3 cm in size, attached to cloudy danburite were found. Chalcopyrite is found in all of the Charcas mines (Minrec 55.6.743-744).
At the Mponeng Mine, West Wits, Far West Rand, West Rand District Municipality, Gauteng, South Africa, brass-coloured crystals of chalcopyrite less than 6 mm have been found included in quartz and on baryte, associated with galena and pyrrhotite (R&M 96.4.222).
At the Palabora Mine, Phalaborwa, Mopani District, Limpopo, South Africa, chalcopyrite occurs in a carbonatite (R&M 92.5.436-437).
At the Mariquita Mine (Sultana Mine), Usagre, Badajoz, Extremadura, Spain, massive chalcopyrite, one of the primary sulphides in the deposit, is associated with tetrahedrite group minerals, pyrite, cinnabar, and occasionally sphalerite and galena (MinRec 55.4.496).
At Malmberget, Gällivare, Norrbotten County, Sweden, chalcopyrite is found in massive form, mixed with bornite, and as simple sphenoidal crystals from 5 mm to 3 cm. Some specimens show coxcomb aggregates of smaller chalcopyrite crystls, while others show large individual crystals perched on matrix or on emerald-green fluorite octohedrons, sometimes with quartz crystals (MinRec 56.5.586).
Chalcopyrite from Malmberget - IMage
At Alderley Edge, Cheshire, England, UK, copper mineralised solutions percolated through porous sandstone and deposited barium, cobalt, copper, lead, vanadium and zinc minerals between the sand grains. Anhydrite formed as cement in permeable rocks, then baryte was deposited, followed by pyrite, chalcopyrite, sphalerite and galena. Subsequently a second generation of baryte and iron-rich calcite followed. These minerals crystallised from highly saline, sulphate-rich brines, at a temperature of 50 to 60o C (RES pps 49-50). A specimen of chrysocolla with a pseudomorph of goethite after chalcopyrite has been found (RES p55).
At Redruth, Cornwall, England, UK, chalcopyrite has been found with tetrahedrite and calcite (FM 47545).
At the Clargillhead vein, Garrigill, Alston Moor, Eden, Cumbria, England, UK, chalcopyrite is a common sulphide. In addition to its presence as inclusions in galena, it infills void spaces within radiating quartz and appears to be free of inclusions generally, though rare masses of pyrite and gersdorffite up to 5 microns across were found within it. The chalcopyrite has been found intergrown with galena, or altered to chalcocite and covellite, or to banded limonite, sometimes intergrown with small native silver droplets (JRS 23.49).
At Croft quarry, Blaby, Leicestershire, England, UK, chalcopyrite has been found as inclusions in analcime and also disseminated in the host diorite. Chalcopyrite crystals to 1 mm have also been found associated with fluorite, calcite and analcime (JRS 20.13-14, RES 189).
At Breedon quarry, Breedon on the Hill, Leicestershire, England, UK, chalcopyrite occurs with calcite (RES p202, 208).
At Cloud Hill Quarry, Breedon on the Hill, Leicestershire, England, UK, chalcopyrite occurs with calcite, dolomite and baryte (RES p205)
At Earl Ferrers' mine, Staunton Harold, Leicestershire, England, UK, chalcopyrite occurs with galena, sphalerite, calcite, dolomite and baryte (RES p222, 223).
At Barrasford Quarry, Chollerton, Northumberland, England, UK, a few small patches of chalcopyrite have been found within masses of sphalerite within calcite veins JRS 21.8).
At Wotherton mine, Chirbury, Shropshire, England, UK, chalcopyrite occurs with calcite and pyrite (RES p285).
At the Shadwell quarry, Much Wenlock, Shropshire, England, UK, chalcopyrite occurs with calcite on limestone (RES p296).
At the Pennerley mine, near Worthen, Shropshire, England, UK, chalcopyrite occurs with calcite, quartz and baryte (RES p290).
At the Ecton mine, Staffordshire, England, UK, chalcopyrite occurs with calcite, baryte, pyrite, galena and malachite (RES p302-307).
At Judkins quarry, Nuneaton, Warwickshire, England, UK, chalcopyrite has been found as pseudomorphs after chalcocite, with calcite (RES p324)
At the Magma mine, Pioneer District, Pinal county, Arizona, USA, chalcopyrite crystals have been collected with a purple bornite surface, and also on quartz crystals; crystalline masses of chalcopyrite have been found in association with sphalerite (R&M 95.1.84).
At Joplin, Jasper county, Missouri, USA, tetragonal crystals of chalcopyrite have been found epitaxial on sphalerite (FM 47452).
At the Sweetwater Mine, Ellington, Reynolds county, Missouri, USA, chalcopyrite pseudomorphs after galena have been found (KL p131).
Also epimorphs (? perimorphs) of chalcopyrite and pyrite after anhydrite crystals have been found as brilliant pyrite-gold, faintly iridescent spherules and curving tubular aggregates composed of densely packed chalcopyrite and/or pyrite microcrystals enclosing long, flat, hollow spaces once occupied by bladed crystals of what was probably anhydrite (Minrec 55.1.82).
At the Cooney Mine, Mogollon Mining District, Mogollon Mountains, Catron County, New Mexico, USA, iridescent chalcopyrite has been found with quartz (Mindat photo).
Chalcopyrite from the Cooney Mine - Image
At Balmat, Fowler, St. Lawrence County, New York, USA, chalcopyrite occurs as masses and crystals in small quantities in all of the Balmat mines. Lustrous sphenoidal chalcopyrite crystals to 2 cm were found at the #3 mine, sometimes studding the surfaces of calcite crystals. Botryoidal masses of chalcopyrite were also reported from the 900-foot level of the #3 mine and at the #4 mine (MinRec 56.1.44).
At the Bingham Canyon Mine, Bingham Canyon, Bingham Mining District, Salt Lake County, Utah, USA, chalcopyrite has been found in quartz monzonite porphyry together with well-formed mirror-bright blades of molybdenite (Mindat photo).
Chalcopyrite from the Bingham Canyon Mine - Image
At the Luck Fairfax Plant, Loudoun county, Virginia, USA, chalcopyrite is found embedded in green prehnite, occasionally partially altered to bornite (R&M 98.2.126).
At the Luck Leesburg Plant, Leesburg, Loudoun county, Virginia, USA, sharp chalcopyrite crystals to about 5 mm have been found, accompanied by epidote and white stilbite crystals (R&M 98.2.126).
At the Vulcan Materials Company Crushed Stone Quarry, Manassas, Prince William county, Virginia, USA, a 1.9-cm chalcopyrite crystal on green prehnite has been found (R&M 98.2.126).
At the Mufulira Mine, Mufulira, Mufulira District, Copperbelt Province, Zambia, chalcopyrite, like bornite and chalcocite, forms dense disseminations along bedding planes and is less common in veins, where it is associated with bornite and chalcocite and commonly with quartz. Well formed crystals are uncommon but occasionally occur in vugs and quartz veins in the orebodies (MinRec 55.4.456-461).
Chalcopyrite from Mufulira - Image
Alteration
chalcopyrite to goethite
The first stage of copper supergene mineral formation is the oxidation of a primary copper-containing ore mineral, such as chalcopyrite. This is a complex process, but the reaction can be summarised as:
chalcopyrite + oxygen + water → copper ions + sulphate ions + goethite + sulphuric acid
4CuFeS2(s) + 17O2(g) + 6H2O(l) → 4Cu2+(aq) + 4SO42-(aq) + 4FeO(OH)(s) + 4H2SO4(aq)
The copper and sulphate ions are then free to react with carbonate ions in solution to form a range of supergene copper sulphates and carbonates (JRS 18.12).
chalcopyrite to sulphur
Oxidation of pyrite forms ferrous (divalent) sulphate and sulphuric acid:
pyrite + oxygen + water → ferric sulphate + sulphuric acid
FeS2 + 7O + H2O → FeSO4 + H2SO4
The ferrous (divalent) sulphate readily oxidizes to ferric (trivalent) sulphate and ferric hydroxide:
ferrous sulphate + oxygen + water → ferric sulphate + ferric hydroxide
6FeSO4 + 3O + 3H2O → 2Fe2(SO4)3 + 2Fe(OH)3
Ferric sulfate is a strong oxidizing agent; it oxidises chalcopyrite according to the reaction:
chalcopyrite and ferric sulphate to copper sulphate, ferrous sulphate and sulphur
CuFeS2 + 2Fe2(SO4)3 → CuSO4 + 5FeSO4 + 2S
(AMU b3-3.7)
chalcopyrite, arsenopyrite, CO2 and O2 to Fe-tennantite, siderite and sulphur
10CuFeS2 + 4FeAsS + 4CO2 + 8O2 = Cu10Fe2As4S13 + 4Fe(CO3) + 11/2S2
(CM 28.725-738)
chalcopyrite, arsenopyrite and pyrite to Fe-tennantite and troilite
10CuFeS2 + 4FeAsS + FeS2 = Cu10Fe2As4S13 + 13FeS
(CM 28.725-738)
chalcopyrite, arsenopyrite and sulphur to Fe-tennantite and pyrite
10CuFeS2 + 4FeAsS + 13/2S2 = Cu10Fe2As4S13 + 12FeS2
This reaction occurs at a comparatively low temperature (CM 28.725-738).
chalcopyrite, arsenopyrite and sulphur to Fe-tennantite and troilite
l0CuFeS2 + 4FeAsS + l/2S2 = Cu10Fe2As4S13 + l2FeS
(CM 28.725-738)
chalcopyrite and chalcocite to bornite
CuFe3+S2 + 2Cu2S = Cu5FeS4
This reaction occurs in the supergene enrichment zone (JRS 18.14).
chalcopyrite, stibnite and sulphur to Fe-tetrahedrite and pyrite
10 CuFeS2 + 2 Sb2S3 + 3/2 S2 = Cu10Fe2As4S13 + 8FeS2
(CM 28.725-738)
enargite and pyrite to Fe-tennantite, chalcopyrite and sulphur
4Cu3AsS4 + 4FeS2 = Cu10Fe2As4S13 + 2CuFeS2 + 7/2S2
(CM 28.725-738)
Fe-tetrahedrite, berthierite and sulphur to chalcopyrite and stibnite
Cu10Fe2Sb4S13 + 2FeSb2S4 + 11/2S2 = 10CuFeS2 + 4Sb2S3
(CM 28.725-738)
Fe-tetrahedrite, siderite and sulphur to chalcopyrite, stibnite, CO2 and O2
Cu10Fe2Sb4S13 + 8Fe(CO3) + 13/2S2 = 10CuFeS2 + 2Sb2S3 + 8CO2 + 4O2
(CM 28.725-738)
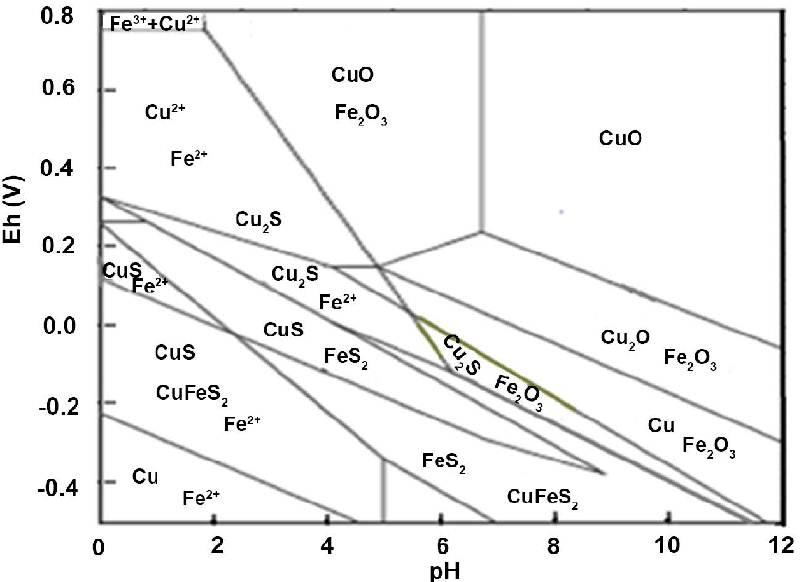
The diagram below is a Pourbaix diagram for Cu-Fe-S-H2O (IJNM 07(02).9.23). It shows the relationship between copper Cu, chalcopyrite CuFeS2, tenorite CuO, covellite CuS, cuprite Cu2O, chalcocite Cu2S, pyrite FeS2 and hematite Fe2O3.
Back to Minerals