It is frequently found as pseudomorphs after azurite, or as alteration pseudomorphs after cuprite. It occurs less frequently as pseudomorphs after atacamite, brochantite, chalcopyrite, tetrahedrite, chalcophyllite, gypsum, libethenite, calcite, sphalerite, cerussite, and pyrite. It is found rarely altered to azurite or cuprite (Mindat).
Malachite is a relatively high pH (alkaline) mineral, and brochantite converts to malachite as the pH increases. If the carbonate content of the environment increases, then the boundary where malachite is more stable than brochantite moves to a lower pH (more acid) environment (JRS 18.13).
Localities
The Two Mile and Three Mile deposits, Paddy's River, Paddys River District, Australian Capital Territory, Australia, are skarn deposits at the contact between granodiorite and volcanic rocks. malachite is a secondary carbonate occurring as crusts and fibres on magnetite and actinolite (AJM 22.1.42).
At the Mount Kelly deposit, Gunpowder District, Queensland, Australia, the copper ores overlie primary zone mineralisation consisting of quartz-dolomite-sulphide veins hosted in siltstone and schist. Malachite is the most common secondary copper mineral in the deposit. Overgrowths and coatings of malachite were observed on azurite, chrysocolla, romanèchite and hematite (AJM 22.1.23).
At the Sir Dominick Mine, Yudnamutana District, Arkaroola Region, North Flinders Ranges, Flinders Ranges, South Australia, the copper mineralisation lies within steeply dipping shear zones in metamorphic rocks and consists entirely of azurite, cuprite and malachite; no sulphides have been encountered at this deposit. Malachite pseudomorphs after azurite occur as single crystals, rosette groups and spherical aggregates. Some of the pseudomorphs retain what appears to be a core of original azurite. The deposit is hosted by a sequence of carbonate-deficient rocks in a region that experiences very low rainfall (R&M 97.6.534-555).
Malachite from the Sir Dominick Mine - Image
At the Ibiajara district, Rio do Pires, Bahia, Brazil, some fine malachite pseudomorphs after azurite have been found, comprising multiple rosette-like sheaves of tabular azurite crystals, completely altered to a vibrant velvety-green malachite; resting aesthetically on these replacements were spherules of lighter green, non-pseudomorphous malachite (R&M 97.6.534-555).
Malachite from Ibiajara - Image
At the Shilu Mine, Yangchun County, Yangjiang, Guangdong, China, masses of silky emerald-green malachite are not uncommon (AESS).
Malachite from Shilu - Image
At the Lin Ma Hang mine, North District, New Territories, Hong Kong, China, the lead-zinc deposit is a hydrothermal deposit which lies along a fault zone within altered acid volcanic rocks, consisting mainly of chlorite, biotite, sericite and actinolite, with scattered quartz. (Hong Kong Minerals (1991). Peng, C J. Hong Kong Urban Council)
The mineralisation consists of a series of fissure vein deposits varying from a few mm to several metres in width. The initial vein filling was coarse milky quartz. this was followed by an intrusion of fine-grained quartz carrying the metallic minerals, galena, pyrite, sphalerite and chalcopyrite, in order of abundance (Geological Society of Hong Kong Newsletter, 9.4.3-27).
Malachite and azurite are occasionally found in close association with chalcopyrite (Hong Kong Minerals (1991). Peng, C J. Hong Kong Urban Council).
Malachite from Lin Ma Hang - Image
The Ma On Shan Mine, Ma On Shan, Sha Tin District, New Territories, Hong Kong, China, is an abandoned iron mine, with both underground and open cast workings. The iron ores contain magnetite as the ore mineral and occur predominantly as masses of all sizes enclosed in a large skarn body formed by contact metasomatism of dolomitic limestone at the margins of a granite intrusion. In parts of the underground workings magnetite is also found in marble in contact with the granite. The skarn rocks consist mainly of tremolite, actinolite, diopside and garnet. Malachite and azurite are occasionally found in close association with chalcopyrite, formed by the action of carbonated water on the chalcopyrite (Hong Kong Minerals (1991). Peng, C J. Hong Kong Urban Council)
The Lin Fa Shan deposit, Tsuen Wan District, New Territories, Hong Kong, China, is located in a remote area of the Tai Mo Shan Country Park, on a steep west facing slope of Lin Fa Shan, just above the abandoned village of Sheung Tong. The surrounding hillsides are covered with shallow excavations, representing past searches for wolframite, the natural ore of tungsten. The abandoned workings are extremely dangerous with unsupported tunnels, open shafts and no maintenance since their closures in 1957; the workings should not be entered (http://industrialhistoryhk.org/lin-shan).
Malachite and azurite are occasionally found in close association with chalcopyrite formed by the action of carbonated waters on the chalcopyrite (Hong Kong Minerals (1991). Peng, C J. Hong Kong Urban Council).
At the Zhaojue copper mine, Sikai, Zhaojue County, Liangshan Yi, Sichuan, China, malachite occurs as crusts on sandstone (AESS).
Malachite from Zhaojue - Image
At the Mindingi Mine, Kambove Territory, Haut-Katanga, DR Congo, an odd-looking sort of malachite was found in the late spring/early summer of 2023. The very dark (because goethite-infused) malachite forms globular aggregates built of tightly packed acicular crystals, and the globules rest lightly on matrix plates of sparkling, microcrystallised quartz Minrec 55.1.112-113).
Malachite from Mindingi - Image
At the Shangulowe mine, Kambove district, Democratic Republic of Congo, malachite pseudomorphs after baryte have been found (KL p180).
At Chessy-les-Mines, Villefranche, Rhône, Auvergne-Rhône-Alpes, France, malachite pseudomorphs after azurite have been found (R&M 95.3.275).
Malachite from Chessy - Image
At Rudabánya, Kazincbarcika District, Borsod-Abaúj-Zemplén County, Hungary, in 1985 a spectacular matrix specimen was found consisting of a group of sharp, deep blue crystals of azurite, with parts of the crystals altered to bright green malachite in a limonite cavity. The largest crystal in the group is 16 cm long and 8 cm wide while the others are 10 to 12 cm long (R&M 97.6.534-555).
Malachite from Rudabánya - Image
At the Nakhlak Mine, Anarak District, Nain County, Isfahan Province, Iran, epigenetic (formed later than the surrounding or underlying rock formation) vein deposits and metasomatic replacement bodies are hosted by a chalky Upper Cretaceous (100.5 to 66 million years ago) limestone. The limestone underwent dolomitisation prior to sulphide mineralisation. The principal primary ore mineral is galena, associated with minor or trace amounts of sphalerite, tetrahedrite -tennantite, pyrite and chalcopyrite as inclusions. The main secondary ore mineral is cerussite, sometimes associated with minor amounts of anglesite, plattnerite, wulfenite, minium, mimetite, covellite, chalcanthite, malachite and goethite. Many trace elements are present in the primary galena, but most notably it is rich in silver and antimony and poor in bismuth.
Malachite occurs very rarely and in small quantities, as an alteration product of chalcopyrite. Botryoidal velvet malachite with cerussite crystals has been collected here (Minrec 54.3.383-408).
At Dzezkazgan, Kazakstan, malachite has been found with iodargyrite (FM 42699).
The Khanong open pit, Sepon Mine, Vilabouly District, Savannakhet Province, Laos, exploited a large deposit of supergene chalcocite in contact with limestone and dolomitic country rocks. The best azurite and malachite specimens were found beneath the chalcocite orebody where acidic, copper-bearing groundwater that had percolated through the sulphide ore reacted with the carbonate country rock. Some pristine azurites with no malachite alteration were found; others were completely altered to malachite. The vast majority of Sepon azurites, however, showed first generation crystals completely replaced by malachite but then apparently overgrown with a very thin, incomplete layer (typically less than 1 mm) of fresh, highly lustrous azurite (R&M 97.6.534-555).
Malachite from Khanong - Image
At the Ojuela Mine, Mapimí, Mapimí Municipality, Durango, Mexico, malachite pseudomorphs after thin, transparent bladed azurite crystals to 1 cm on a gossan matrix have been found (R&M 97.6.534-555).
Malachite from Ojuela - Image
At Kerrouchen Aït Ikaiss, El Kbab Cercle, Khénifra Province, Béni Mellal-Khénifra Region, Morocco, fine vibrant blue azurite specimens and pseudomorphs of malachite after azurite have been collected. They were extracted from small cavities, up to 10 cm across, in dolostone. The locality is equally well known for cone-shaped sprays of tightly packed, radiating acicular malachite (R&M 97.6.534-555).
Malachite from Kerrouchen - Image
At Touissit-Bou Beker mining district, Jerada Province, Oriental Region, Morocco, the deposit is a Mississippi Valley–type (MVT) lead-zinc deposit, hosted in calcareous shale, sandstone, limestone and dolostone. Galena and sphalerite are the primary ore minerals found in dolostone voids. The presence of copper in MVT deposits is unusual but not unknown; where it does occur, copper is much less prevalent than lead and zinc, as it is here.
Late-stage dissolution and oxidation produced solution cavities that host collectible minerals, including very sharp pseudomorphs of malachite after prismatic azurite, with a velvet luster, up to 20 cm in size (R&M 97.6.534-555).
Malachite from Touissit - Image
The Milpillas Mine, Cuitaca, Santa Cruz Municipality, Sonora, Mexico, produced world-class specimens of malachite pseudomorphs after azurite. Malachite is the stable species under normal atmospheric carbon dioxide levels and azurite requires slightly elevated levels and slightly more acidic conditions than malachite. Milpillas originated as a low-grade, intrusive-hosted chalcopyrite-bornite-pyrite porphyry copper deposit which was subjected to 40 million years of erosion and ground-water infiltration that gradually oxidised and upgraded the low-grade primary ore to a high-grade supergene deposit. Many finds from the mine feature malachite pseudomorphs after azurite on the same specimen with completely unaltered azurite crystals; “electric-blue” azurite has a layer of pristine azurite over a pseudomorphs of malachite after azurite; in other finds malachite pseudomorphs after azurite to 5 cm long were associated with vésigniéite, and separately occurred on an iridescent goethite matrix (R&M 97.6.534-555).
Malachite from Milpillas - Image
At the Aranzazú Mine, Concepción del Oro, Concepción del Oro Municipality, Zacatecas, Mexico, fine malachite pseudomorphs after azurite occur. The most outstanding examples are robust sharp, isolated, elongate chisel-tipped crystals to perhaps 4 cm with roughly equant cross-sections (R&M 97.6.534-555).
Malachite from Aranzazú - Image
At the Onganja mining area, Seeis, Windhoek Rural, Khomas Region, Namibia, rare prismatic malachite pseudomorphs after azurite crystals up to 10 cm in size have been found. The copper ore consists predominantly of chalcocite, chalcopyrite, malachite, cuprite and native copper (R&M 97.6.534-555).
Malachite from Onganja - Image
At Tsumeb, Oshikoto Region, Namibia, malachite occurs as pseudomorphs after azurite and, rarely, after cuprite (R&M 93.6.545). Also rosasite pseudomorphs after malachite after azurite with cerussite have been found (KL p181).
Malachite is the most abundant secondary copper mineral at Tsumeb. Non-pseudomorphous (so-called “primary”) malachite crystals occur rarely as blocky crystals to 1 cm and more abundantly as small needles and crusts of “velvet” malachite, sometimes with cuprite and native copper. A few of the blocky “primary” crystals have also been found in the third oxidation zone on Level 43. The most celebrated specimens are the large and attractive pseudomorphs and partial pseudomorphs after azurite and other minerals which reach many centimeters in size. At least 30 other minerals are known to be sometimes associated, including duftite, mimetite, mottramite and smithsonite, as well as those listed above (Minrec 55.6 supplement p110).
Malachite from Tsumeb - Image
At the Delfina Mine, Hoyos de Alda, Ortiguero, Prado, Cabrales, Asturias, Spain, twisted malachite has been found (Mindat photo).
Malachite from the Delfina Mine - Image
At the Mariquita Mine (Sultana Mine), Usagre, Badajoz, Extremadura, Spain, although malachite is a common secondary mineral, it does not form significant specimens; usually it appears as small masses and pulverulent crusts. Occasionally it is seen as radial groups of acicular crystals to 1 cm in length, and sometimes, when associated with the yellow mercury oxychlorides, calomel, zálesíite and rosasite, it forms beautiful colour-contrasting micromount specimens (MinRec 55.4.504).
Malachite from Mariquita - Image
At Malmberget, Gällivare, Norrbotten County, Sweden, green malachite stains and small encrustations are sometimes seen on specimens of copper, chalcopyrite and bornite (MinRec 56.5.596).
Malachite from Malmberget - Image
At Alderley Edge, Cheshire, England, UK, supergene azurite and malachite are common (RES pps 49-50), and cuproasbolane has been found associated with malachite (RES p53).
At Balliway Rigg, Caldbeck, Allerdale, Cumbria, England, UK, a 4 cm specimen of iron stained quartz matrix with a rich cover, front and back, of fibrous malachite and minor chrysocolla has been found (AESS).
Malachite from Balliway Rigg - Image
At Red Gill Mine, Roughton Gill, Caldbeck, Allerdale, Cumbria, England, UK, a specimen was found with vugs containing a combination of malachite, cerussite and brochantite crystals with light blue chrysocolla (AESS).
Malachite from Red Gill - Image
At Roughton Gill Mine, Roughton Gill, Caldbeck, Allerdale, Cumbria, England, UK, microscopic green, vitreous, acicular crystals of malachite occur on and in quartz (AESS).
Malachite from the Roughton Gill Mine - Image
At the Brundholme Mine, Glenderaterra Valley, Threlkeld, Eden, Cumbria, England, UK, malachite crystals have been found on chrysocolla from this very rare British locality, together with pyrite or chalcopyrite and other unidentified minerals (AESS).
Malachite from the Brundholme Mine - Image
At the Snelston mine, near Ashbourne, Derbyshire, England, UK, malachite occurs on sandstone (RES p140).
Malachite from Snelston - Image
At Croft Quarry, Croft, Blaby, Leicestershire, England, UK, very small spherules and encrustations of malachite are associated with oxidised chalcopyrite; the presence of such green oxidation products can be useful in distinguishing chalcopyrite from marcasite. (JRS 20.20-21).
At Lane's Hill quarry, Stoney Stanton, Blaby, Leicestershire, England, UK, malachite occurred intimately associated with djurleite in a large vein of Fe-bearing dolomite. Present usually as thin films and encrustations, it occasionally occurred as minute single crystals, up to 1.1 mm in length (JRS 20.21).
At Bardon Hill quarry, Coalville, Leicestershire, England, UK, malachite occurs with azurite on dacite (RES p193).
Malachite from Bardon Hill - Image
At Newhurst quarry, Shepshed, Leicestershire, England, UK, malachite has been found with minor baryte, replacing earlier chalcopyrite and bornite (RES p199).
Malachite from Newhurst - Image
At Breedon quarry, Breedon on the Hill, Leicestershire, England, UK, malachite has been found with calcite (RES p203).
At the Eardiston mine, near West Felton, Shropshire, England, UK, malachite occurs on sandstone (RES p291).
At Llynclys quarry, near Oswestry, Shropshire, England, UK, malachite occurs with chalcopyrite, goethite and dolomite (RES p294, 295).
Malachite from Llynclys - Image
At Judkins quarry, Nuneaton, Warwickshire, England, UK, malachite is associated with calcite (RES p324).
At Bisbee, Cochise county, Arizona, USA, fine pseudomorphs of malachite after azurite on limonitic matrix have been found in many mines, including the Campbell, Cole, Sacramento and Junction mines (R&M 94.2.167, KL p179).
Malachite from Bisbee - Image
At the Live Oak Pit of the Inspiration mine, Gila county, Arizona, USA, coatings of chalcedony over chrysocolla form over malachite replacements of azurite. Also many specimens of malachite replacing azurite, some perched on chrysocolla, have come from this locality (R&M 94.2.162).
Malachite from the Live Oak Pit - Image
At the Morenci mining district, Greenlee County, Arizona, USA, azurite and malachite production was from limestone-hosted oxidised ores. Fine malachite pseudomorphs after azurite were recovered that typically exhibit silky, chatoyant surface features and commonly occur as blocky groups, rosettes, or single crystals. Many show a second generation of azurite growth, and most are partially altered by late-stage weathering. They are associated with azurite, chrysocolla, goethite and black manganese oxides on a distinctive siliceous quartz-rich matrix (R&M 97.6.534-555).
Malachite from Morenci - Image
At the New Cornelia Mine, Ajo, Little Ajo Mountains, Ajo Mining District, Pima county, Arizona, USA, beautiful malachite pseudomorphs after azurite nearly always occur as roughly equant, blocky crystals, either as singles or lumpy aggregates on manganese-oxide-coated porphyry host rock. Most have a rough yet somewhat velvety surface, but some have smooth striated faces. Many of the specimens exhibit azurite crystals completely altered to malachite and juxtaposed with unaltered azurite crystals (R&M 97.6.534-555).
Malachite from the New Cornelia Mine - Image
At the Ray mine, Pinal county, Arizona, USA, malachite pseudomorphs after gypsum have been found (R&M 94.2.165).
Malachite from the Ray Mine - Image
At the Mammoth-Saint Anthony Mine, St. Anthony deposit, Tiger, Mammoth Mining District, Pinal county, Arizona, USA, many fine specimens of malachite pseudomorphs after azurite have been recovered, ranging from slender elongated replacements to blocky crystals to 6 cm in length. Tiger malachite tends to be highly chatoyant, with broken surfaces revealing long fibres of malachite or patchworks of fibres that grew during the replacement process. Some of the best-known Tiger examples are radial aggregates of slender azurite crystals entirely replaced by malachite. These commonly show a second stage of azurite growth, generally parallel to the original azurite. Malachite pseudomorphs after azurite from Tiger nearly always occur with cerussite (R&M 97.6.534-555).
Malachite from the Mammoth-Saint Anthony Mine - Image
At the Bagdad mine, Yavapai county, Arizona, USA, rare pseudomorphs of malachite after azurite have been found (R&M 94.2.164). The crystals tend to be stellate medium green sprays on a brick-red to light tan matrix. The individual crystals are typically flattened, thin and elongated, often radiating outward from a central nucleation point. In many instances the weathering process was prolonged, resulting in pseudomorphs of chrysocolla after malachite after azurite, but these chrysocolla pseudomorphs tend to be unstable and may desiccate, crack, and change colour through dehydration and continued oxidation.
Malachite from the Bagdad Mine - Image
At the Piedmont mine, Yavapai county, Arizona, USA, extremely rare fine specimens of centimetre sized pseudomorphs of malachite after azurite have been found, coated with a crust of quartz (R&M 94.2.167-168).
Malachite from the Piedmont Mine - Image
At the Mohawk Mine, Mohawk Hill, Clark Mountain District, Clark Mts, San Bernardino County, California, USA, malachite has been found with goethite and cuprite (Mindat photo)<\i>.
Malachite from the Mohawk Mine - Image
At the G-2 Prospect, Bete Grise, Grant Township, Keweenaw County, Michigan, USA, malachite has been found with adularia and chalcocite (Mindat photo).
Malachite from the G-2 Prospect - Image
At the Tintic Mining District, Juab County, Utah, USA, malachite is a common secondary mineral in the copper-rich areas such as Mammoth. A variety of styles are seen from acicular malachite to botryoidal masses and a variety of pseudomorphs (MinRec 55.2.209).
Malachite from Tintic - Image
At the Apex mine, Jarvis Peak, Beaver Dam mountains, Washington county, Utah, USA, an azurite and malachite pseudomorph after gypsum has been found (KL p177).
Malachite from the Apex Mine - Image
At the Kabwe mine, Central Province, Zambia, malachite is an extremely rare secondary copper mineral, but it has been found as a coating on, and partly replacing, cerussite. Also in silicified dolomite, with a mammillary habit, with malachite at the core, passing through zinc-rich malachite to rosasite in the outer layer (R&M 94.2.130).
Malachite from Kabwe - Image
At the Mufulira Mine, Mufulira, Mufulira District, Copperbelt Province, Zambia, malachite is the most common secondary copper mineral. It often occurs as an oxidation product of primary chalcopyrite grains or blebs and is widespread in both the near-surface oxidised zone and in deeper oxide zones in the orebodies. Three generations of malachite have been observed, the first two of which make for collectible mineral specimens:
(a) as pseudomorphous replacements of probable azurite, in the form of large divergent sprays of tabular crystals
(b) as secondary overgrowths of divergent acicular crystals to 5 mm in cavities in oxide zones, intimately associated with massive cuprite
(c) as a late-stage near-surface alteration product of chalcopyrite and bornite grains, seen as small irregular patches and powdery coatings, sometimes with a remnant core of the original sulphide (MinRec 55.4.472-474).
Alteration
azurite and H2O to malachite and CO2
2Cu3(CO3)2(OH)2 + H2O → 3Cu2(CO3(OH)2 + CO2
Azurite is unstable under atmospheric conditions, and slowly converts to the more stable malachite according to the above reaction. This instability is evidenced by the existence of many pseudomorphs of malachite after azurite; pseudomorphs of azurite after malachite are extremely rare (MM 50.41-47).
duftite (s) and H2CO3 (aq) to cerussite (s), malachite (s), H2AsO4- (aq) and H+ (aq)
2PbCuAsO4(OH) + 3H2CO3 ⇌ 2PbCO3 + Cu2CO3(OH)2 + 2H2AsO4- + 2H+
(MM 52.688)
The Activity-pH diagram below was calculated at 298.2 K for some carbonates and copper arsenates for constant activity (roughly equivalent to concentration) of H2AsO4- in solution, over a range of values of pH and of H2CO3 activity (MM 52.687).
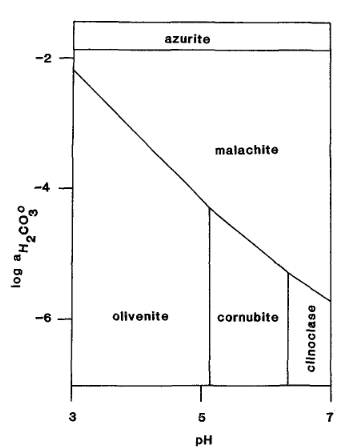
The mineral formulae are:
azurite: Cu3(CO3)2(OH)2
malachite: Cu2(CO3)(OH)2
olivenite: Cu2(AsO4)(OH)
cornubite: Cu5(AsO4)2(OH)4
clinoclase: Cu3(AsO4)(OH)3
Back to Minerals