It often occurs as slender crystals inside quartz and mica. Rutile is found in considerable quantities in black sands associated with ilmenite, magnetite, zircon and monazite.
It is a mineral of the greenschist, amphibolite, granulite and eclogite facies.
Localities
At Carver's Claim, Wadnaminga goldfield, Pastoral Unincorporated Area, South Australia, rutile has been found (Mindat photo)
Rutile from Carver's Claim - Image
At Mount Moliagul, Moliagul, Central Goldfields Shire, Victoria, Australia, small groups of blood-red to brownish rutile crystals have been found in cavities in quartz (AJM 21.1.44).
At the Mount Deverell variscite deposit, Milgun Station, Western Australia, rutile occurs in the country rock and also is included in veins of variscite that have been replaced by foggite or crandallite. Also as inclusions in quartz. The variscite deposits are hosted by marine sedimentary rocks (AJM 20.2.29).
At Novo Horizonte, Bahia, Brazil, fine specimens of rutile with hematite have been found (Mindat photo).
Rutile from Novo Horizonte - Image
At the Santa Rosa pegmatite district, Itambacuri, Minas Gerais, Brazil, rutile, probably containing some tantalum (variety strüverite) or niobium (variety ilmenorutile), is known from the Santa Rosa Field as lustrous, blocky to thick bladed black crystals to 2.5 cm in size (MinRec 56.4.453)
At the Monte Cervandone area, Devero Alp, Baceno, Verbano-Cusio-Ossola Province, Piedmont, Italy, Rutile is very common in the gneisses around Cervandone, although it is seldom seen in fine specimens. A feature of the region is niobium-rich rutile, often found with the arsenic minerals of the clefts. Niobium-rich rutile occurs in a range of colours from yellow-brown through red-brown and almost black, in skeletal aggregates and as masses of fibrous crystals replacing anatase. Lattice-like reticulated formations of rutile also occur in clefts in the gneisses, and Wannigletscher collecting sites have given up druses of brilliant red, short-prismatic microcrystals of rutile accompanying cervandonite and synchysite (MinRec 56.3.318).
Rutile from the Monte Cervandone area - Image
At Magnet Cove, Arkansas, USA, pseudomorphs of rutile after brookite have been found (KL p140).
The Ryerson Hill Quarry, Paris, Oxford County, Maine, USA, is in a complex pegmatite. Rutile was found in the core zone between two previously excavated pits where smoky quartz and hydroxlherderite were found. Rutile occurs as straw-colored aggregates ranging from 1 to 3 mm in size and is associated with clear to brownish gemmy microsized tourmaline rods; it is found near the contact of the pegmatite with mica schist (R&M 97.5.449).
Alteration
Hydrothermal alteration of titanium-bearing minerals commonly releases titanium, resulting in the formation of rutile or anatase (AofA). Rutile is the principle alteration mineral of ilmenite; it can replace ilmenite, and may in turn be associated with later growth crystals of ilmenite (R&M 16.2.100, AJM 18.2.26).
amphibole, chlorite, paragonite, ilmenite, quartz and calcite to garnet, omphacite, rutile, H2O and CO2
NaCa2(Fe2Mg3)(AlSi7)O22(OH)2 + Mg5Al(AlSi3O10)(OH)8 + 3NaAl2(Si3Al)O10(OH)2 + 4Fe2+Ti4+O3 + 9SiO2 + 4CaCO3 → 2(CaMg2Fe3)Al4(SiO4)6 + 4NaCaMgAl(Si2O6)2 + 4TiO2 + 8H2O + 4CO2
In low-grade rocks relatively rich in calcite the garnet-omphacite association may be due to reactions such as the above (DHZ 2A p453).
amphibole, clinozoisite, chlorite, albite, ilmenite and quartz to garnet, omphacite, rutile and H2O
NaCa2(Fe2Mg3)(AlSi7)O22(OH)2 + 2Ca2Al3[Si2o7][SiO4]O(OH) + Mg5Al(AlSi3O10)(OH)8 + 3NaAlSi3O8 + 4Fe2+Ti4+O3 + 3SiO2 → 2(CaMg2Fe3)Al4(SiO4)6 + 4NaCaMgAl(Si2O6)2 + 4TiO2 + 6H2O
In low-grade rocks relatively poor in calcite the garnet-omphacite association may be developed by the above reaction (DHZ 2A p453).
augite, albite, pyroxene, anorthite and ilmenite to omphacite, garnet, quartz and rutile
2MgFe2+Si2O6 + Na(AlSi3O8) + Ca2Mg2Fe2+Fe3+AlSi5O18 + 2Ca(Al2Si2O8) + 2Fe2+Ti4+O3 → NaCa2MgFe2+Al(Si2O6)3 + (Ca2Mg3Fe2+4)(Fe3+Al5)(SiO4)9 + SiO2 + 2TiO2
This reaction occurs at high temperature and pressure (DHZ 2A p449).
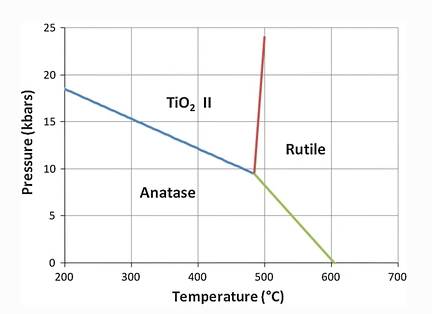
The PT diagram below illustrates that rutile is stable at a higher temperature than anatase ( Journal of Materials Science, 46.855–874).
Back to Minerals