Localities
At the type locality, Lavrion slag localities, Lavreotiki, East Attica, Attica, Greece, paralaurionite was found in ancient lead slags altered by seawater (Dana) and associated with laurionite, penfieldite, fiedlerite and phosgenite (HOM).
Paralaurionite from the Lavrion slag localities - Image
At Toussit, Oujda, northern Morocco, paralaurionite occurs as large bladed crystals with leadhillite (Dana).
At Shepton Mallet, Mendip Hills, Somerset, England, UK, paralaurionite is common in large cleavage masses in hydrocerussite (Dana). It is also reported as being locally common as yellow patches in mendipite or the calcite - cerussite cavity infill, or as colourless, white or greyish thin tablets between mendipite cleavages (JRS 13.24).
At the Mammoth-Saint Anthony mine, Saint Anthony deposit, Tiger, Mammoth Mining District, Pinal county, Arizona, USA, paralaurionite is associated with leadhillite, matlockite, cerussite, hydrocerussite, diaboleite and wherryite (HOM).
Paralaurionite from the Mammoth-Saint Anthony Mine - Image
Stability
The Activity-pH diagram below was calculated for some lead minerals. Boundaries are calculated for constant activity (roughly equivalent to concentration) of (SO4)2- and constant partial pressure (also roughly equivalent to concentration) of CO2, over a range of values of pH and of Cl1- activity. In this case the concentration of CO2 is above the atmospheric value.
Paralaurionite and phosgenite can co-exist at this CO2 concentration. At higher concentrations phosgenite is the stable mineral, and at lower concentrations paralaurionite is the stable mineral. If the concentration of CO2 decreases further the stability field of paralaurionite extends to include an environment which is more acid (lower pH) and has a lower Cl- activity (JRS 15.22).
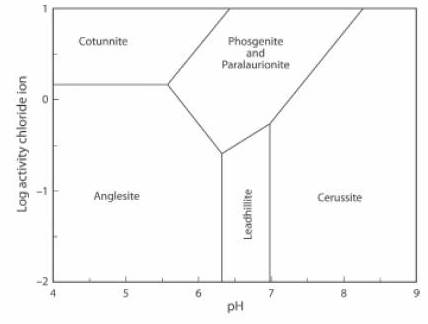
The lead mineral formulae are:
cotunnite PbCl2
phosgenite Pb2(CO3)Cl2
paralaurionite PbCl(OH)
cerussite Pb(CO3)
anglesite Pb(SO4)
leadhillite Pb4(CO3)2(OH)2
The Activity-pH diagram below is similar, but the concentration of CO2 is close to zero, at about 0.01% of the atmospheric value, and the (SO4)2- activity is about 0.5% of its value in the first diagram (JRS 15.18-23).
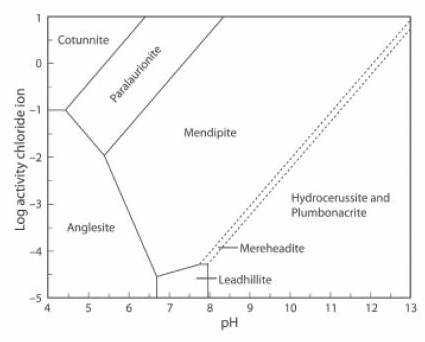
The lead mineral formulae are:
cotunnite PbCl2
paralaurionite PbCl(OH)
mendipite Pb3O2Cl2
mereheadite Pb47O24(OH)13cl25(BO3)2(CO3)
hydrocerussite Pb3(CO3)2(OH)2
plumbonacrite Pb5(CO3)3O(OH)2
anglesite Pb(SO4)
leadhillite Pb4(CO3)2(OH)2
Back to Minerals