Localities
At the type locality, Torr Works Quarry (Merehead Quarry), Cranmore, Mendip, Somerset, England, UK, mereheadite is associated with mendipite, blixite, cerussite, hydrocerussite and calcite in lenses and pods within veins of manganese and iron oxide minerals which cut dolomitic limestone (AM 84.1196).
Mereheadite is found as massive patches of orange-brown to dark brown granular material completely filling carbonate lined cavities in manganese oxides, or as isolated regions entirely within mendipite. A number of mereheadite specimens contain areas of massive but glassy dark orange material, which is also pure mereheadite. Some of the white material found on Merehead quarry specimens in 2005 was also found to be mereheadite. The orange colour variation might be tied both to a change in boron content and to a slight change in size of the unit cell; darker specimens have slightly more borate and a slightly smaller unit cell size (JRS 13.26).
Stability
The Activity-pH diagram below was calculated for some lead minerals. Boundaries are calculated for constant activity (roughly equivalent to concentration) of (SO4)2- and constant partial pressure (also roughly equivalent to concentration) of CO2, over a range of values of pH and of Cl1- activity. Here the concentration of CO2 is close to zero, at about 0.01% of the atmospheric value
Cerussite does not form in these conditions, the stability field of mendipite is very large, and mereheadite and plumbonacrite can form, although they are not stable at higher levels of concentration of CO2 (JRS 15.18-23).
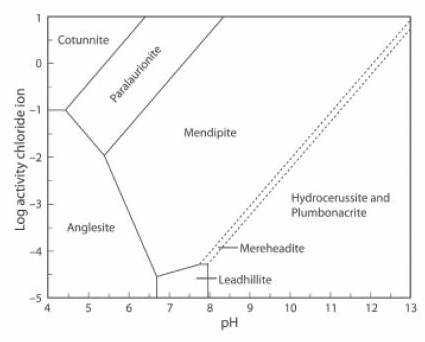
The lead mineral formulae are:
cotunnite PbCl2
paralaurionite PbCl(OH)
mendipite Pb3O2Cl2
mereheadite Pb47O24(OH)13cl25(BO3)2(CO3)
hydrocerussite Pb3(CO3)2(OH)2
plumbonacrite Pb5(CO3)3O(OH)2
anglesite Pb(SO4)
leadhillite Pb4(CO3)2(OH)2
Back to Minerals