At the type locality, Churchill, Mendip, Somerset, England, UK, mendipite occurs in nodules in manganese oxide ores, associated with hydrocerussite, cerussite, malachite, pyromorphite, calcite, chloroxiphite, diaboleite and parkinsonite (HOM, Mindat). Mendipite forms only when the supply of CO2 is restricted; if it is not, cerussite forms instead. This is why mendipite can be found only in the sealed environment of a cavity in the manganese oxides, isolated from the surrounding limestone which otherwise would be a source of abundant CO2, since the Mendips is a range of limestone hills (JRS 13.23).
Mendipite from Mendip - Image
Alteration
The mendipite - hydrocerussite equilibrium reaction is
mendipite + CO2 + water ⇌ hydrocerussite + H+ + Cl-
Pb3O2Cl2 + 2CO2 + 2H2O ⇌ Pb3(CO3)2(OH)2 + 2H+ + 2Cl-
(JRS 15.27).
In addition to only forming at relatively high pH (alkaline), mendipite requires a steady supply of a moderately strong base to provide the hydroxyl ions (OH-) needed for the reaction.
3Pb2+ + 4OH- + 2Cl- = Pb3O2Cl2 (mendipite) + 2H2O
(JRS 15.25).
If there is an ample supply of litharge, mendipite can form according to the reaction
litharge + Cl- + water = mendipite + (OH)-
3PbO + 2Cl- + H2O = Pb3O2Cl2 + 2(OH)-
(JRS 15.26).
Stability
The Activity-pH diagram below was calculated for some lead minerals. Boundaries are calculated for constant activity (roughly equivalent to concentration) of (SO4)2- and constant partial pressure (also roughly equivalent to concentration) of CO2, over a range of values of pH and of Cl1- activity. In this case the concentration of CO2 is approximately equal to the atmospheric value.
Mendipite has a small stability field here, although it is unstable at higher CO2 concentrations. If the concentration of CO2 decreases further the stability field of mendipite extends to include a very wide range of pH and of Cl- activity.
For seawater aCl- = 10-0.5 and pH = 8.2. Here these values are outside the stability field of mendipite, so it cannot form in seawater. As the concentration of CO2 decreases, however, the stability field of mendipite widens until it includes these values, and the mineral can form in seawater (JRS 15.18-23).
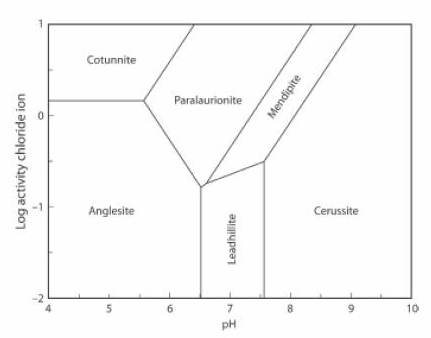
The lead mineral formulae are:
cotunnite PbCl2
paralaurionite PbCl(OH)
mendipite Pb3O2Cl2
cerussite Pb(CO3)
anglesite Pb(SO4)
leadhillite Pb4(CO3)2(OH)2
The Activity-pH diagram below is similar, but the concentration of CO2 is close to zero, at about 0.01% of the atmospheric value, and the (SO4)2- activity is about 0.5% of its value in the first diagram.
Cerussite does not form in these conditions, the stability field of mendipite is very large, and mereheadite and plumbonacrite can form, although they are not stable at higher levels of concentration of CO2 (JRS 15.18-23).
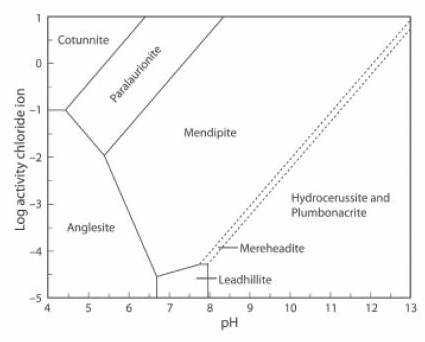
The lead mineral formulae are:
cotunnite PbCl2
paralaurionite PbCl(OH)
mendipite Pb3O2Cl2
mereheadite Pb47O24(OH)13cl25(BO3)2(CO3)
hydrocerussite Pb3(CO3)2(OH)2
plumbonacrite Pb5(CO3)3O(OH)2
anglesite Pb(SO4)
leadhillite Pb4(CO3)2(OH)2
Back to Minerals