Localities
At Caracoles, Chile, cotunnite is associated with galena, cerussite, anglesite and matlockite (HOM).
At the type locality, Mount Vesuvius, Somma-Vesuvius Complex, Naples, Campania, Italy, cotunnite occurs in fumeroles (Dana).
At the Tolbachik volcano, Kamchatka Krai, Russia, cotunnite is associated with tenorite, ponomarevite, sofiite, burnsite, ilinskite, georgbokiite, chloromenite, halite, sylvite and gold (HOM).
At the Torr Works Quarry (Merehead Quarry), Cranmore, Mendip, Somerset, England, UK, cotunnite has been found on one specimen in very small amounts as minute grains associated with massive diaboleite. The presence of cotunnite indicates a pH of 3 was present, at least locally. These extremely acid conditions represent the final stages of mineral formation (JRS 13.32).
At Penrhyn Du Mine, Abersoch, Llanengan, Lleyn Peninsula, Gwynedd, Wales, UK, cotunnite has been found as crystals to 4 mm in a cavity in oxidised galena, associated with a few tiny sulphur crystals. The specimen was collected from a vein exposed below the High Water mark. It is likely that a reaction between chlorine in the seawater and lead in galena in the veinstone produced this rare mineral (MW)
Stability
The Activity-pH diagram below was calculated for some lead minerals. Boundaries are calculated for constant activity (roughly equivalent to concentration) of (SO4)2- and constant partial pressure (also roughly equivalent to concentration) of CO2, over a range of values of pH and of Cl1- activity. In this case the concentration of CO2 is appreciably more than the atmospheric value.
Cotunnite is stable in an acid environment with a high concentration of Cl- ions (JRS 15.18-23).

The lead mineral formulae are:
cotunnite PbCl2
phosgenite Pb2(CO3)Cl2
cerussite Pb(CO3)
anglesite Pb(SO4)
The Activity-pH diagram below is similar, but in this case the concentration of CO2 is close to zero, at about 0.01% of the atmospheric value, and the (SO4)2- activity about 0.5% of its value in the first diagram (JRS 15.18-23).
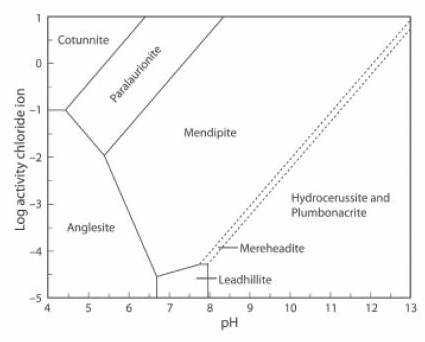
The lead mineral formulae are:
cotunnite PbCl2
paralaurionite PbCl(OH)
mendipite Pb3O2Cl2
mereheadite Pb47O24(OH)13cl25(BO3)2(CO3)
hydrocerussite Pb3(CO3)2(OH)2
plumbonacrite Pb5(CO3)3O(OH)2
anglesite Pb(SO4)
leadhillite Pb4(CO3)2(OH)2
Back to Minerals