In Tsumeb, Namibia, hydrocerussite may be of primary origin, but usually it is a secondary mineral, formed by the alteration of cerussite. This requires highly alkaline conditions with a pH of 10 to 13, and a constant and stable concentration of carbonate ions in solution. These conditions are most likely only at depth, and indeed a deep pocket has been found which yielded twinned cerussite partially or wholly replaced by hydrocerussite, associated with litharge, massicot, minium and scotlandite, all occurring as microcrystals on the hydrocerussite. Other associated minerals include dundasite, galena, smithsonite, mottramite, dioptase, plancheite, cobaltoan dolomite, tennantite and wulfenite. Hydrocerussite from Tsumeb rarely forms as pseudomorphs after anglesite.
Tsumeb hydrocerussite has been found from three different mineral assemblages. Firstly scaly aggregates associated with azurite, malachite, mimetite and calcite (the hydrocerussite occurs only on the azurite). Secondly intergrown with or overgrown on cerussite, associated with arsentsumebite, azurite, malachite, mimetite and phosgenite. Lastly as pseudomorphs of hydrocerussite after cerussite; some of these crystals still contain unaltered cores, and these are associated with mimetite, duftite and dolomite (R&M 93.2.150-151, KL p178).
At the type locality, Långban, Sweden, hydrocerussite is most commonly found as an alteration product of native lead, associated with litharge, hausmannite or clinopyroxene skarn (R&M 93.2.155.).
At the Higher Pitts Mine, Mendip Hills, England, UK, hydrocerussite is fairly common as massive nodules, and also forms as an alteration product around nodules of mendipite (R&M 93.2.153-154).
At the Potter-Cramer Mine, Vulture Mining District, Maricopa county, Arizona, USA, hydrocerussite occurs as a secondary mineral forming thin white crusts on cerussite, which is an alteration product of galena (R&M 96.1.32).
At the Mammoth-Saint Anthony mine, Tiger, Pinal county, Arizona, USA, hydrocerussite is associated with diaboleite, caledonite, paralaurionite and cerussite (R&M 93.2.155).
Alteration
For carbonate concentration slightly lower than atmospheric, cerussite and hydrocerussite can co-exist in alkaline environments, with leadhillite in more acid environments, and anglesite in yet more acid environments. For lower carbonate concentrations hydrocerussite completely replaces cerussite. In these conditions anglesite and hydrocerussite cannot co-exist (JRS 18.11).
Litharge, CO2 and H2O to hydrocerussite
3PbO + 2CO2 + H2O → Pb3(CO3)2(OH)2
Synthetic hydrocerussite can be easily obtained, as a white powder, by the action of carbon dioxide and water on litharge at pH 4 to 5 (R&M 93.2.150).
The mendipite-hydrocerussite equilibrium reaction is
mendipite + CO2 + water ⇌ hydrocerussite + H+ + Cl-
Pb3O2Cl2 + 2CO2 + 2H2O ⇌ Pb3(CO3)2(OH)2 + 2H+ + 2Cl-
(JRS 15.27)
An ample supply of CO2 favours the forward reaction.
Stability
The Activity-pH diagram below was calculated for some lead minerals. Boundaries are calculated for constant activity (roughly equivalent to concentration) of (SO4)2- and constant partial pressure (also roughly equivalent to concentration) of CO2, over a range of values of pH and of Cl1- activity. In this case the concentration of CO2 is less than the atmospheric value.
Hydrocerussite and cerussite can co-exist at this level of CO2 concentrations. At higher concentrations cerussite is the stable mineral, and at lower concentrations hydrocerussite is the stable mineral (JRS 15.18-23).
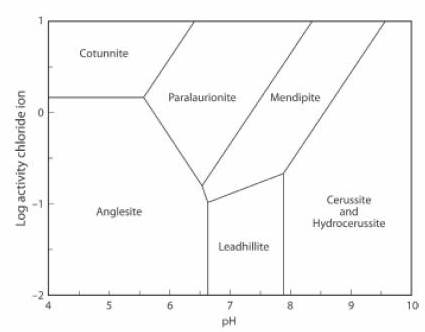
The lead mineral formulae are:
cotunnite PbCl2
paralaurionite PbCl(OH)
mendipite Pb3O2Cl2
cerussite Pb(CO3)
hydrocerussite Pb3(CO3)2(OH)2
anglesite Pb(SO4)
leadhillite Pb4(CO3)2(OH)2
The Activity-pH diagram below is similar, but the concentration of CO2 is close to zero, at about 0.01% of the atmospheric value, and the (SO4)2- activity is about 0.5% of its value in the first diagram.
Hydrocerussite and plumbonacrite can co-exist here. At lower CO2 concentration plumbonacrite is the stable mineral, and at higher concentration the stable mineral is hydrocerussite, then, at an even higher concentration, cerussite (JRS 15.18-23).
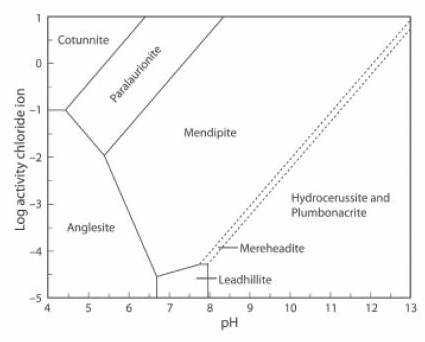
The lead mineral formulae are:
cotunnite PbCl2
paralaurionite PbCl(OH)
mendipite Pb3O2Cl2
mereheadite Pb47O24(OH)13cl25(BO3)2(CO3)
hydrocerussite Pb3(CO3)2(OH)2
plumbonacrite Pb5(CO3)3O(OH)2
anglesite Pb(SO4)
leadhillite Pb4(CO3)2(OH)2
Back to Minerals