Localities
At the type locality, the Chañarcillo Mining District, Copiapó, Copiapó Province, Atacama, Chile, adamite is associated with silver, limonite and chlorargyrite (Mindat).
Adamite from the Chañarcillo Mining District - Image
At Johanngeorgenstadt, Erzgebirgskreis, Saxony, Germany, in 2005 a surprising find of adamite was reported in transparent, colourless to white spheres with a radial structure, perched on white carbonate rock interspersed with pale brown sphalerite (MinRec 55.5.587).
At the Hilarion Mine, Kamariza Mines, Agios Konstantinos, Lavrion Mining District, Lavreotiki, East Attica, Attica, Greece, adamite has been found associated with hydrozincite (Mindat photo).
Adamite from the Hilarion Mine - Image
At Tsumeb, Namibia, adamite is associated with smithsonite, goethite and zincolivenite (R&M 95.3.210-232).
Adamite from Tsumeb - Image
At the Caldbeck Fells Mining Region, Cumbria, England, UK, adamite is rare. At the Deer Hills baryte mine it is associated with agardite-mixite and aurichalcite. At the Driggith mine cuprian adamite has been found encrusting quartz. At Old Potts Gill mine it occurs with serpierite or malachite, and at Sandbeds mine it occurs as crystals or coatings on quartz (C&S).
Adamite is abundant and widespread at the San Rafael Mine, Nevada, USA. It usually occurs alone there, but it has been found associated with wulfenite and smithsonite, or with segnitite and duftite (R&M.85.6.514-515).
Adamite from San Rafael - Image
Alteration
legrandite to adamite and H2O
Zn2AsO4(OH). H2O (s) ⇌ Zn2AsO4(OH) (s) + H20(l)
Legrandite is a very much rarer mineral than adamite and it is suggested that it is in fact metastable with respect to adamite at room temperature, although it may crystallise as the stable phase under appropriate conditions at temperatures between 0 and 25oC. It is of interest that legrandite has been found at several localities without adamite (MM 52.685).
The Activity-pH diagram below was calculated at 298.2 K for some zinc arsenates and Ca3(AsO4)2 for constant activity (roughly equivalent to concentration) of Ca2+ ions in solution, over a range of values of pH and of Zn2+ activity (MM 52.685).

The zinc mineral formulae are:
adamite: Zn2(AsO4)(OH)
köttigite: Zn3(AsO4)2.8H2O
austinite: CaZn(AsO4)(OH)
The Activity-pH diagram below was calculated at 298.2 K for smithsonite, hydrozincite and adamite for constant activity (roughly equivalent to concentration) of H2AsO4- in solution, over a range of values of pH and of H2CO3 activity (MM 52.688).
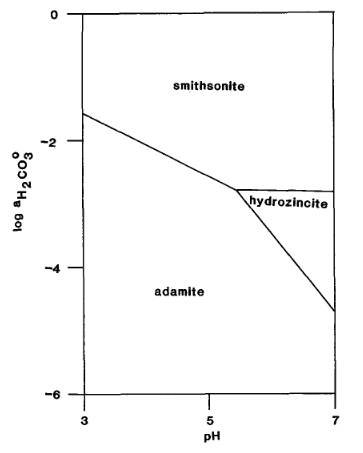
The mineral formulae are:
smithsonite: Zn(CO3)
hydrozincite: Zn5(CO3)2(OH)6
adamite: Zn2(AsO4)(OH)
Back to Minerals